6-K
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 6-K
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16 UNDER
THE SECURITIES EXCHANGE ACT OF 1934
For the month of January 2026
Commission File Number: 001-39173
NovaBridge Biosciences
2440 Research Boulevard, Suite 400
Rockville, MD 20850
(Address of principal executive offices)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F.
Form 20-F ☒ Form 40-F ☐
On January 6, 2026, NovaBridge Biosciences issued a press release and an investor presentation, copies of which are furnished herewith as Exhibit 99.1 and Exhibit 99.2, respectively.
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
|
|
|
|
|
NovaBridge Biosciences |
|
|
|
By |
: |
/s/ Xi-Yong Fu |
|
Name |
: |
Xi-Yong (Sean) Fu |
|
Title |
: |
Chief Executive Officer |
Date: January 6, 2026
EX-99.1

NovaBridge Presents Positive Givastomig Dose Expansion Data from the Phase 1b Combination Study in Patients with 1L Metastatic Gastric Cancer
•Givastomig, a CLDN18.2 x 4-1BB bispecific antibody, continues to show robust efficacy when combined with nivolumab and chemotherapy (mFOLFOX6) in 1L HER2-negative, metastatic gastric cancer patients, with 77% ORR observed at 8 mg/kg and 73% ORR observed at 12 mg/kg, across a wide range of PD-L1 and CLDN18.2 expression levels
•The median PFS was 16.9 months at 8 mg/kg; 12 mg/kg is immature with approximately 4-month shorter median follow-up; data will be updated in 2026
•Six-month landmark PFS was 73% for 8 mg/kg, and 91% for 12 mg/kg cohorts
•Combination was well tolerated; safety is comparable to the current standard of care treatment
•Data demonstrate that givastomig is a potential best-in-class CLDN18.2 asset when added to 1L standard of care
•NovaBridge is on track to initiate enrollment in a global, randomized Phase 2 study, evaluating both doses against standard of care, in Q1 2026
•Detailed Phase 1b dose expansion data are expected to be presented at a medical conference later in 2026
ROCKVILLE, MD, January 6, 2026 – NovaBridge Biosciences (Nasdaq: NBP) (NovaBridge or the Company) a global biotechnology platform company committed to accelerating access to innovative medicines, today announced positive updated results from the Phase 1b combination study of givastomig, a Claudin 18.2 (CLDN18.2) x 4-1BB bispecific antibody, in combination with nivolumab and chemotherapy (mFOLFOX6) in patients with HER2-negative, first line (1L) metastatic gastric cancer.
The results combine data from patients in the Phase 1b dose escalation and expansion cohorts treated with 8 mg/kg or 12 mg/kg of givastomig. An ORR of 77% (20/26) at the 8 mg/kg dose, and 73% (19/26) at the 12 mg/kg dose, confirms and extends positive signals observed in the dose escalation cohorts. Responses continue to be rapid and deepen over time, and were observed across all levels of CLDN18.2 and PD-L1 expression levels. The safety profile of the combination was similar to earlier observations, except for the emergence of immune-related gastritis, which correlated with improved clinical outcomes. These data, outlined in detail below, position givastomig as a potential best-in-class CLDN18.2-directed therapy for gastric cancer, a potential $12 billion market opportunity by 2030. The full data are intended to be presented at a medical meeting later in 2026.
The Phase 1b study (NCT04900818) is evaluating the safety, efficacy, pharmacokinetics (PK), and pharmacodynamics (PD) of givastomig, used in combination with nivolumab and mFOLFOX6, as 1L therapy in patients with CLDN18.2-positive gastric cancer (GC) (≥1+ immunohistochemistry (IHC) intensity in ≥1% of cells), and any level of PD-L1 expression. The primary endpoint is safety. Secondary endpoints include progression free survival (PFS). The study enrolled only patients in the U.S.
“The dose expansion data reinforce the strong signals we observed in dose escalation. The efficacy is clear at 8 mg/kg, with robust ORRs, including in subgroups defined by low PD-L1 and/or CLDN18.2 expression. The PFS data are very promising and continue to mature. Emerging efficacy data at 12 mg/kg are also strong and similar in terms of ORR. The 12 mg/kg cohort was enrolled after the 8 mg/kg cohort, so follow-up is shorter and PFS is less mature. We expect to report these data later in 2026. We remain enthusiastic about the 12 mg/kg dose because exposure analysis shows higher
exposure is consistently associated with a higher probability of response, without a higher probability of toxicity,” said Phillip Dennis, MD, PhD, Chief Medical Officer of NovaBridge.
“I continue to be encouraged by givastomig’s high response rate across a wide range of Claudin 18.2 and PD-L1 expression levels, and the depth and duration of responses achieved with combination therapy. The development of gastritis was not predicted by the monotherapy study, and may be related to longer givastomig exposure duration, something that has been seen with some CLDN18.2-directed agents. Most gastritis cases were low grade and manageable, and interestingly appear to be associated with higher response rates and longer survival,” said Samuel J. Klempner, MD, Associate Professor of Medicine at Massachusetts General Hospital. “Givastomig’s favorable benefit-risk balance has the potential to offer real world benefit to patients and is planned to be investigated in a randomized trial.”
“Givastomig is a core program for NovaBridge,” said Sean Fu, PhD, MBA, Chief Executive Officer of NovaBridge. “The compelling Phase 1b data presented today have the potential to establish givastomig as the leading CLDN18.2-directed therapy for 1L gastric cancer, where the unmet medical need remains high and the commercial opportunity is significant.”
Topline Data from the Givastomig Phase 1b Dose Escalation and Expansion Combination Study in 1L Gastric Cancer
•54 advanced metastatic gastric cancer patients (metastatic gastric, esophageal or gastroesophageal adenocarcinomas) were enrolled in cohorts across the 8 mg/kg (n=27), and 12 mg/kg (n=27) dose levels as of the December 2, 2025 data cutoff. 52 patients were efficacy evaluable
•Demographics show characteristics typical of metastatic gastric cancer in patients
•All patients were HER2-negative, CLDN18.2-positive (defined as ≥1+ IHC staining intensity in ≥1% of tumor cells), regardless of PD-L1 expression levels
•Enrolled at sites only within the United States
•Biomarker expression consistent with prevalence, noting that in efficacy evaluable patients:
o8 mg/kg cohort was over-represented with tumors expressing CLDN18.2 >75% (63%)
o12 mg/kg cohort was over-represented with tumors expressing PD-L1 <1% (34%)
Efficacy Results: 51/52 patients exhibited a reduction in the volume of target lesions per RECIST v1.1.
|
|
|
Phase 1b Combination Data Based on patients in the dose escalation and dose expansion cohorts |
Dose level |
8 mg/kg |
12 mg/kg |
Enrolled patients |
27 |
27 |
Efficacy evaluable patients (n)1 |
26 |
26 |
ORR: % (n) |
77 (20/26) |
73 (19/26) |
|
77 (20/26) |
69 (18/26) |
|
- |
4 (1/26) |
|
19 (5/26) |
27 (7/26) |
|
4 (1/26) |
- |
DCR3 % (n) |
96 (25/26) |
100 (26/26) |
Median time to onset of response
(months, Min, Max) |
1.8 (1.3, 7.5) |
2.5 (1.5, 5.4) |
Footnotes:
1. Efficacy evaluable patients defined as having had at least one evaluable on-treatment scan
2. The 12 mg/kg cohort includes three patients with unconfirmed partial responses, still on treatment 3. DCR defined as patients with a complete response (CR), partial response (PR) confirmed and unconfirmed, or stable disease (SD) |
Biomarker Expression
|
Phase 1b ORR Combination Data by Status |
|
|
|
Based on patients in the dose escalation and dose expansion cohorts |
Dose level |
8 mg/kg |
12 mg/kg |
PD-L1 ≥1 |
74 (17/23) |
75 (12/16) |
PD-L1 <1 |
100 (3/3) |
70 (7/10) |
|
|
|
CLDN18.2 ≥75% |
76 (13/17) |
67 (8/12) |
CLDN18.2 <75 % |
78 (7/9) |
79 (11/14) |
Note: For patients with low PD-L1 and low CLDN18.2 the ORR was 83% (5/6) |
Durability of Response Data
|
|
|
Phase 1b PFS Combination Data in Efficacy Evaluable Patients Based on patients in the dose escalation and dose expansion cohorts |
Dose level |
8 mg/kg |
12 mg/kg |
Enrolled patients |
27 |
27 |
Efficacy evaluable patients (n) |
26 |
271 |
Median follow-up (month) |
10.7 |
6.8 |
Events n (%) |
12 (46%) |
5 (19%) |
Censored n (%) |
14 (54%) |
22 (81%) |
Median PFS (mo., 95% CI) |
16.9 (6.8, NA) |
7.7 (6.9, NA) |
6-month PFS rate (95% CI) |
73% (51.7, 86.2) |
91% (69.0, 97.7) |
DOR (month, 95% CI) |
15.2 (5.6, NA) |
NA |
Patients remaining on study |
11 |
18 |
Footnotes: 1. The 12 mg/kg cohort includes one additional patient for survival analysis who was ineligible for response analysis |
Safety: Consistent with previously reported results; incidence of TEAE, TRAE and SAE comparable to 1L combinations in relevant GC benchmark studies (CHECKMATE-649 and SPOTLIGHT), with no dose dependence in TRAE
•Common TRAEs (≥15% of patients in either dose) due to any drug were fatigue, nausea, neutropenia, observed in the majority of patients in each cohort. Grade 3 incidence was low in each cohort (listed as a percentage for 8 mg/kg and 12 mg/kg, respectively): fatigue (7%, 11%), nausea (7%, 4%), and neutropenia (26%, 26%)
•Most common givastomig-related TRAEs (>10% of patients in either dose): nausea, vomiting, and fatigue, all of which had a Grade 3 incidence of ≤11%
•Immune-related gastritis was observed in 33% of patients in each cohort (Grade 3 in 4% of patients dosed at 8 mg/kg and 15% of patients dosed at 12 mg/kg):
oGastritis was not observed in the monotherapy Phase 1 study of givastomig and infrequently observed with nivolumab and chemotherapy
oMost commonly occurred after several cycles of therapy, was documented via endoscopy, and was managed with medications and treatment interruption
•Patients developing gastritis were observed to have improved ORR, PFS and OS compared to patients who did not develop gastritis
•Only Grade 4 TRAE was neutropenia (4% at 8 mg/kg and 7% at 12 mg/kg)
•No Grade 5 TRAEs were reported
Business Update with Leerink Partners
Leerink Partners (Leerink) will host a NovaBridge Business Update on Tuesday, January 6, 2026 at 8:30 AM ET. Interested investors should contact their Leerink representative to join.
Business Update with CITIC Securities
CITIC Securities (CITICS) will host a NovaBridge Business Update (in Chinese) on Wednesday, January 7, 2026 at 9:00 AM HKT. Interested investors should contact their CITICS representative to join.
About Givastomig
Givastomig (TJ033721 / ABL111) is a bispecific antibody targeting Claudin 18.2 (CLDN18.2)-positive tumor cells. It conditionally activates T cells through the 4-1BB signaling pathway in the tumor microenvironment where CLDN18.2 is expressed. Givastomig is being developed for potential treatment of gastric cancer and other Claudin 18.2-positive gastrointestinal malignancies. In Phase 1 trials, givastomig has shown promising anti-tumor activity attributable to a potential synergistic effect of proximal interaction between CLDN18.2 on tumor cells and 4-1BB on T cells in the tumor microenvironment, while minimizing toxicities commonly seen with other 4-1BB agents.
Givastomig is being jointly developed through a global partnership with ABL Bio, in which NovaBridge is the lead party and shares worldwide rights, excluding Greater China and South Korea, equally with ABL Bio.
About NovaBridge
NovaBridge is a global biotechnology platform company committed to accelerating access to innovative medicines. The Company combines deep business development expertise with agile translational clinical development to identify, accelerate, and advance breakthrough assets. By bridging science, strategy, and execution, NovaBridge enables transformative therapies to progress rapidly from discovery toward patients in need.
The Company’s differentiated pipeline is led by givastomig, a potential best-in-class, Claudin 18.2 x 4-1BB bispecific antibody, and VIS-101, a second-in-class, potentially best-in-class bifunctional biologic, targeting VEGF-A and ANG2.
Givastomig conditionally activates T cells via the 4-1BB signaling pathway in the tumor microenvironment where Claudin 18.2 is expressed. Givastomig is being developed to treat Claudin 18.2-positive gastric cancer and other gastrointestinal malignancies. The Company is also collaborating with its partner, ABL Bio, for the development of ragistomig, a bispecific antibody integrating PD-L1 as a tumor engager and 4-1BB as a conditional T cell activator, in solid tumors. Additionally, NovaBridge owns worldwide rights outside of China to uliledlimab, an anti-CD73 antibody that targets adenosine-driven immunosuppression in cancer.
VIS-101 targets VEGF-A and ANG-2 to provide more potent and durable treatment benefits for patients with wet age-related macular degeneration (wet AMD) and diabetic macular edema (DME). VIS-101 is currently completing a large, randomized, dose-ranging Phase 2 study for wet AMD. NovaBridge is the majority shareholder of Visara, and Visara controls global rights to VIS-101, outside of Greater China and certain countries in Asia.
For more information, please visit www.novabridge.com and follow us on LinkedIn.
Forward Looking Statements
This announcement contains forward-looking statements. These statements are made under the “safe harbor” provisions of the U.S. Private Securities Litigation Reform Act of 1995. These forward-looking statements can be identified by terminology such as “will”, “expects”, “believes”, “designed to”, “anticipates”, “future”, “intends”, “plans”, “potential”, “estimates”, “confident”, and similar terms or the negative thereof. NovaBridge may also make written or oral forward-looking statements in its periodic reports to the U.S. Securities and Exchange Commission (the SEC), in its annual report to shareholders, in press releases and other written materials and in oral statements made by its officers, directors or employees to third parties. Statements that are not historical facts, including statements about the Company’s beliefs and expectations, are forward-looking statements. Forward-looking statements in this press release include, without limitation, statements regarding: the strategy, clinical development, plans, results, safety and efficacy of givastomig and VIS-101 and its other drug candidates; the strategic and clinical development of NovaBridge’s drug candidates, including givastomig, ragistomig, uliledlimab, and VIS-101; anticipated clinical milestones and results, and related timing. Forward-looking statements involve inherent risks and uncertainties that may cause actual results to differ materially from those contained in these forward-looking statements, including but not limited to the following: the Company’s ability to
demonstrate the safety and efficacy of its drug candidates; the clinical results for its drug candidates, which may or may not support further development or New Drug Application/Biologics License Application (NDA/BLA) approval; the content and timing of decisions made by the relevant regulatory authorities regarding regulatory approval of the Company’s drug candidates; the Company’s ability to achieve commercial success for its drug candidates, if approved; the Company’s ability to obtain and maintain protection of intellectual property for its technology and drugs; the Company’s reliance on third parties to conduct drug development, manufacturing and other services; the Company’s limited operating history and the Company’s ability to obtain additional funding for operations and to complete the development and commercialization of its drug candidates; and those risks more fully discussed in the “Risk Factors” section in the Company’s annual report on Form 20-F filed with the SEC on April 3, 2025 as well as the discussions of potential risks, uncertainties, and other important factors in the Company’s subsequent filings with the SEC. All forward-looking statements are based on information currently available to the Company. The Company undertakes no obligation to publicly update or revise any forward-looking statements, whether as a result of new information, future events, or otherwise, except as may be required by law.
NovaBridge Investor & Media Contacts
PJ Kelleher
LifeSci Advisors
+1-617-430-7579
pkelleher@lifesciadvisors.com
NovaBridge Biosciences
+1-240-745-6330
IR@novabridge.com

Company Presentation January 2026

Disclaimer This presentation has been prepared by NovaBridge Biosciences (the “Company”) solely for informational purposes. Certain of the information included herein was obtained from various sources, including certain third parties, and has not been independently verified by the Company. By viewing or accessing the information contained in this presentation, you hereby acknowledge and agree that no representations, warranties, or undertakings, express or implied, are made by the Company or any of its directors, shareholders, employees, agents, affiliates, advisors, or representatives (the “Company Relevant Persons”), or any sponsor, underwriter, placing agent, financial advisor, capital market intermediary or any of their respective directors, shareholders, employees, agents, affiliates, advisors, or representatives (collectively with the Company Relevant Persons, the “Relevant Persons”) as to, and no reliance should be placed on the truth, accuracy, fairness, completeness, or reasonableness of the information or opinions presented or contained in, and omission from, this presentation. None of the Relevant Persons shall be responsible or liable whatsoever (in negligence or otherwise) for any loss, howsoever arising from any information presented or contained in this presentation or otherwise arising in connection with the presentation, except to the extent required by applicable law. The information presented or contained in this presentation speaks only as of the date hereof and is subject to change without notice. This presentation includes statistical and other industry and market data that we obtained from industry publications and research, surveys, and studies conducted by third parties, and our own estimates of potential market opportunities. All of the market data used in this presentation involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such data. Industry publications and third-party research, surveys and studies generally indicate that their information has been obtained from sources believed to be reliable, although they do not guarantee, and the accuracy or completeness of such information has not been independently verified. Our estimates of the potential market opportunities for our product candidates include several key assumptions based on our industry knowledge, industry publications, third-party research, and other surveys, which may be based on a small sample size and may fail to accurately reflect market opportunities. While we believe that our internal assumptions are reasonable, no independent source has verified such assumptions. We own or have rights to trademarks or trade names that we use in conjunction with the operation of our business and that appear in this presentation. This presentation also contains references to trademarks and trade names belonging to other entities. All rights to the trademarks, copyrights, logos and other intellectual property listed herein belong to their respective owners and our use or display thereof does not imply an affiliation with, or endorsement by, any other entities. This presentation shall not constitute an offer to sell or the solicitation of an offer to buy these securities, nor shall there be any sale of these securities in any state or jurisdiction in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such state or jurisdiction. This presentation contains forward-looking statements. These statements are made under the “safe harbor” provisions of the U.S. Private Securities Litigation Reform Act of 1995. These forward-looking statements can be identified by terminology such as “future”, “promising”, “may”, “plans”, “potential”, “will”, “could position”, “promise”, “advance”, “target”, “design”, “strategy”, “pipeline”, and “project”, and similar terms or the negative thereof. Statements that are not historical facts, including statements about the Company’s beliefs and expectations, are forward-looking statements. The forward-looking statements in this presentation include, without limitation, statements regarding the following: the Company’s pipeline and capital strategy; the design and potential benefits, advantages, promise, attributes, and target usage of givastomig, ragistomig, uliledlimab and VIS-101; the projected development and advancement of the Company’s portfolio and anticipated clinical milestones, results and related timing; the Company’s expectation regarding the potential market opportunity of gastric cancer, pancreatic ductal adenocarcinoma, cholangiocarcinoma, neovascular age-related macular degeneration and diabetic macular edema; the market opportunity and the Company’s potential next steps (including the potential expansion, differentiation, or commercialization) for givastomig, ragistomig, uliledlimab and VIS-101; the Company’s expectations regarding the impact of data from past, ongoing and future studies and trials; the benefits of the Company’s collaboration with development partners; the timing and progress of studies (including with respect to patient enrollment and dosing); the availability of data and information from ongoing studies; and the Company’s expectations regarding its anticipated cash runway and future use of its cash position. These forward-looking statements involve inherent risks and uncertainties that could cause actual results to differ materially from those expressed or implied in such forward-looking statements. These risks and uncertainties include, but are not limited to, the following: the Company’s ability to demonstrate the safety and efficacy of its drug candidates; the clinical results for its drug candidates, which may or may not support further development or new drug application/biologics license application approval; the content and timing of decisions made by the relevant regulatory authorities regarding regulatory approval of the Company’s drug candidates; the Company’s ability to achieve commercial success for its drug candidates, if approved; the Company’s ability to obtain and maintain protection of intellectual property for its technology and drugs; the Company’s reliance on third parties to conduct drug development, manufacturing and other services; the Company’s limited operating history and the Company’s ability to obtain additional funding for operations and to complete the development and commercialization of its drug candidates; and discussions of potential risks, uncertainties, and other important factors in the Company’s most recent annual report on Form 20-F and the Company’s subsequent filings with the U.S. Securities and Exchange Commission (the “SEC”). The Company may also make written or oral forward-looking statements in its periodic reports to the SEC, in its annual report to shareholders, in press releases and other written materials, and in oral statements made by its officers, directors, or employees to third parties. All forward-looking statements are based on information currently available to the Company. The Company undertakes no obligation to publicly update or revise any forward-looking statements, whether as a result of new information, future events, or otherwise, except as may be required by law.

Company Overview & Highlights
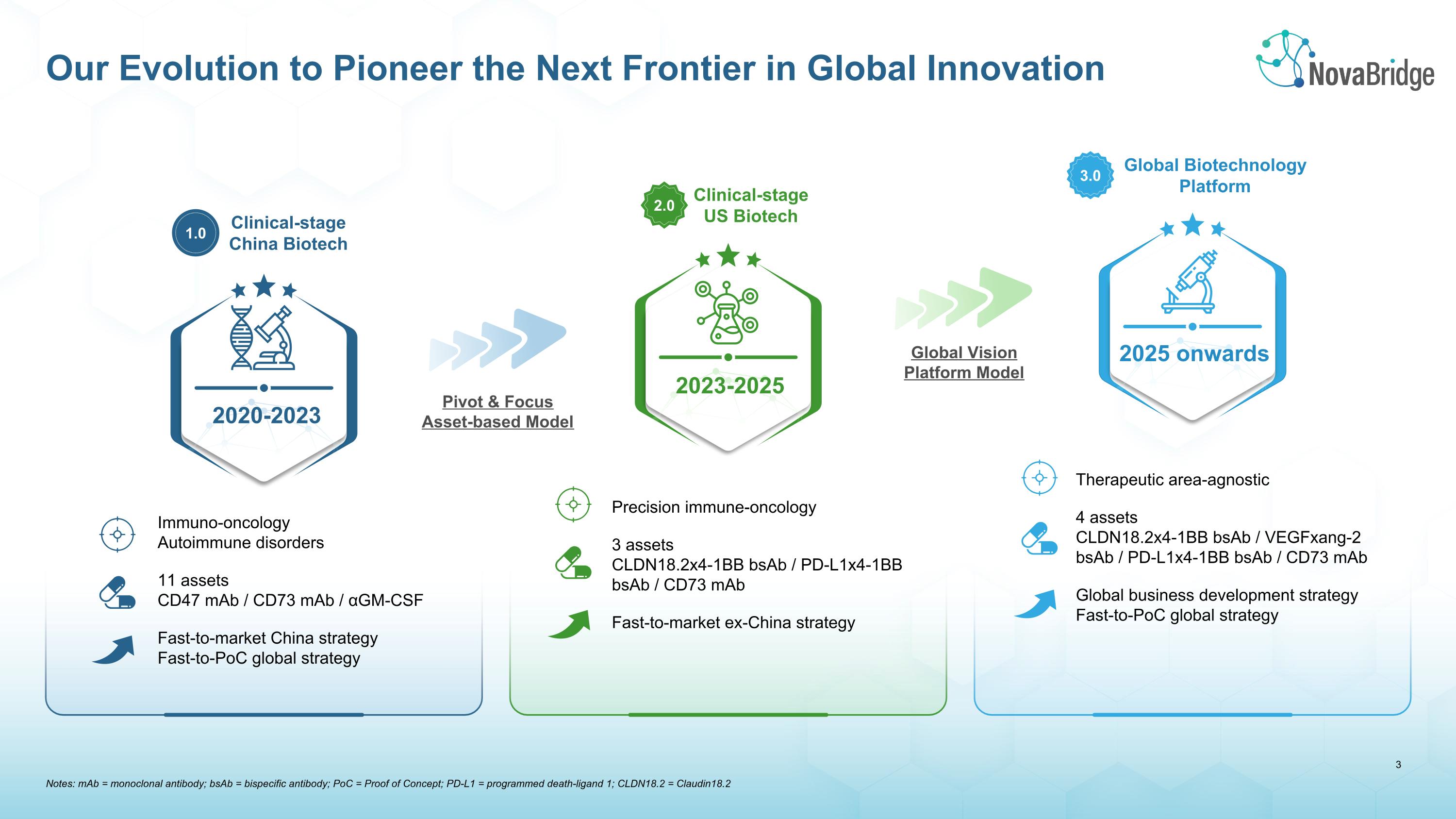
Notes: mAb = monoclonal antibody; bsAb = bispecific antibody; PoC = Proof of Concept; PD-L1 = programmed death-ligand 1; CLDN18.2 = Claudin18.2 Our Evolution to Pioneer the Next Frontier in Global Innovation Pivot & Focus Asset-based Model Clinical-stage �China Biotech 1.0 Clinical-stage �US Biotech 2.0 Global Biotechnology Platform 3.0 Immuno-oncology Autoimmune disorders 11 assets CD47 mAb / CD73 mAb / αGM-CSF Fast-to-market China strategy Fast-to-PoC global strategy Precision immune-oncology 3 assets CLDN18.2x4-1BB bsAb / PD-L1x4-1BB bsAb / CD73 mAb Fast-to-market ex-China strategy Therapeutic area-agnostic 4 assets CLDN18.2x4-1BB bsAb / VEGFxang-2 bsAb / PD-L1x4-1BB bsAb / CD73 mAb Global business development strategy Fast-to-PoC global strategy 2020-2023 Global Vision Platform Model 2023-2025 2025 onwards
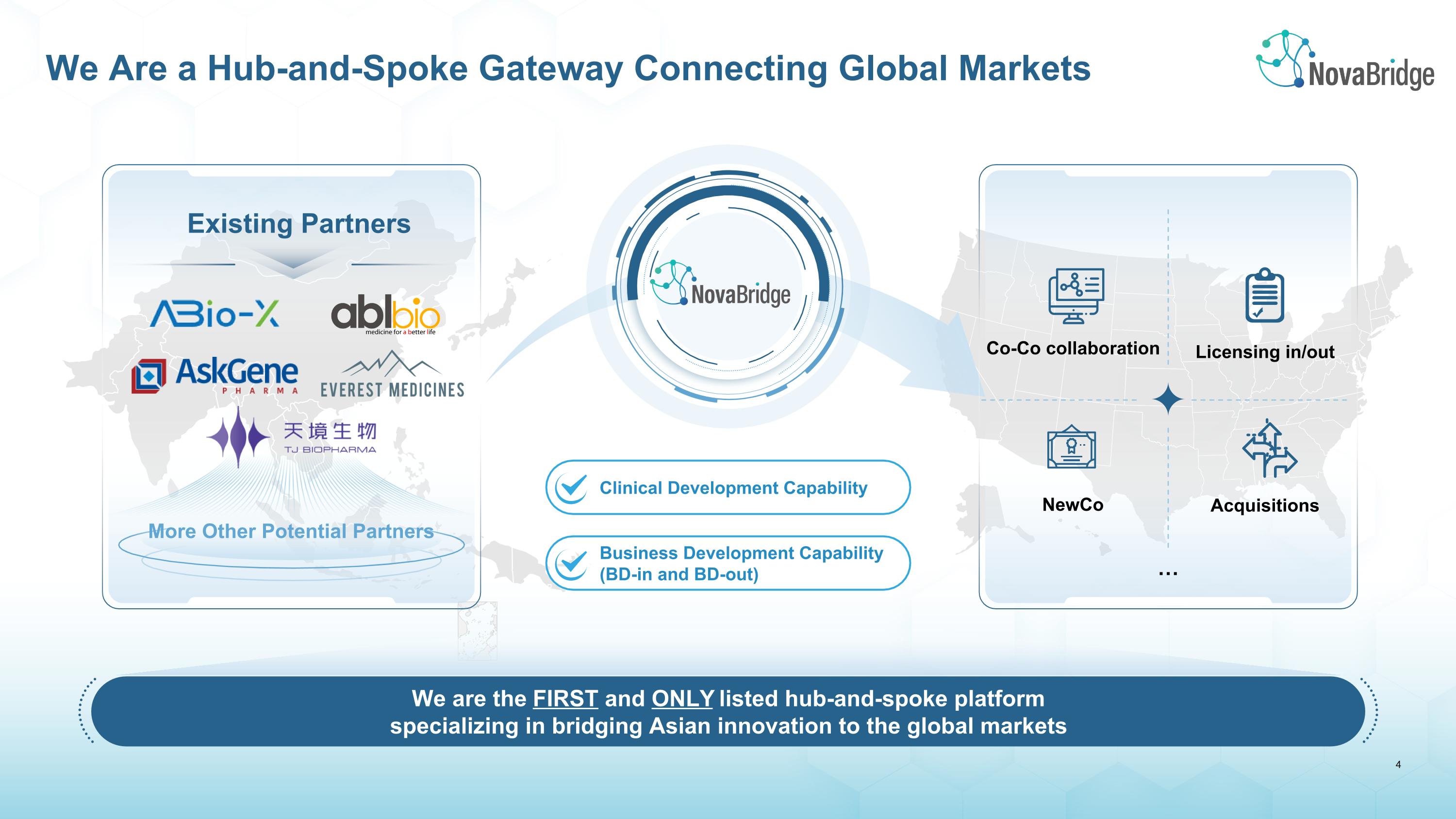
We Are a Hub-and-Spoke Gateway Connecting Global Markets We are the FIRST and ONLY listed hub-and-spoke platform specializing in bridging Asian innovation to the global markets Existing Partners Co-Co collaboration Acquisitions NewCo Licensing in/out … Business Development Capability (BD-in and BD-out) Clinical Development Capability More Other Potential Partners
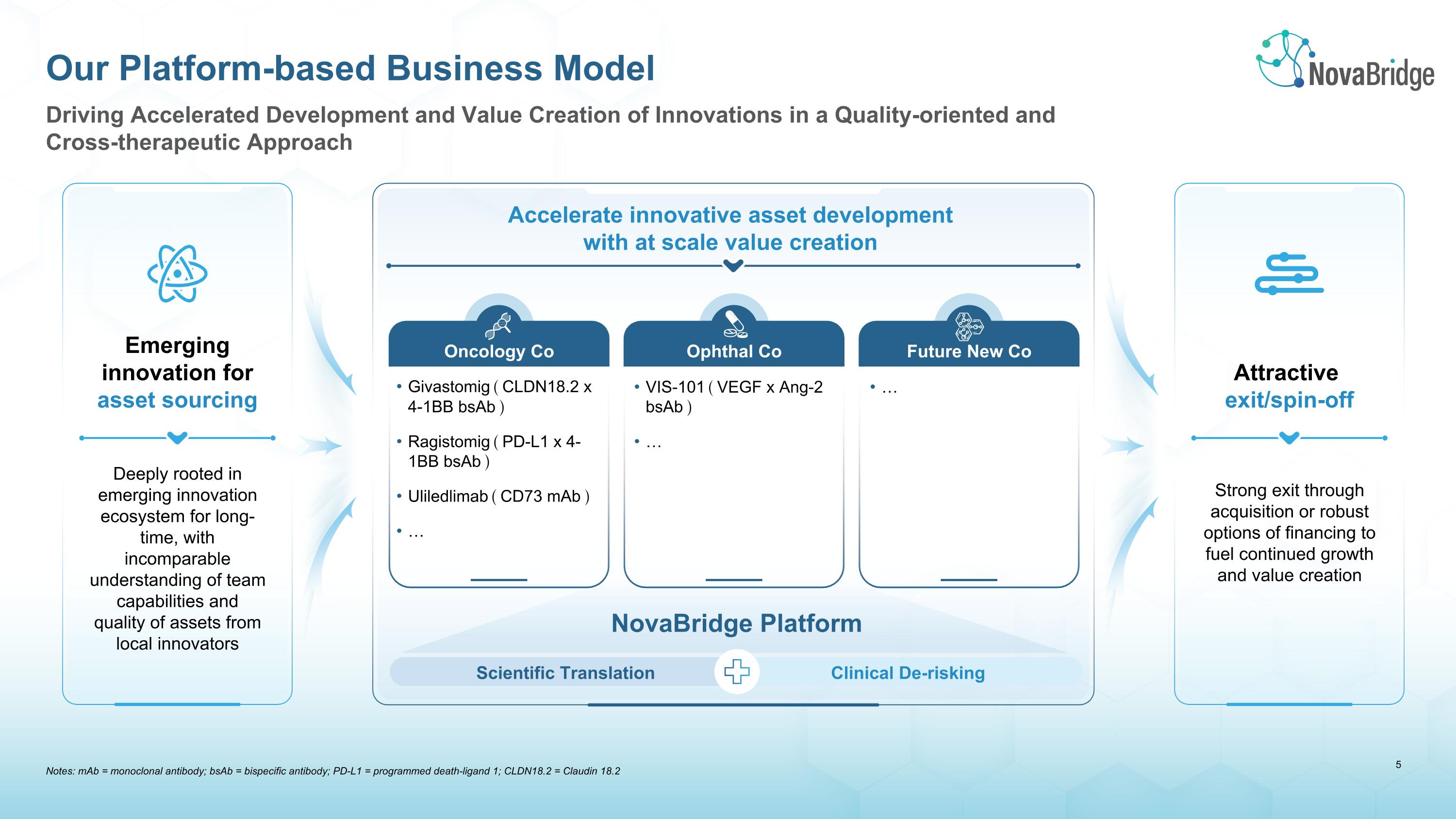
Notes: mAb = monoclonal antibody; bsAb = bispecific antibody; PD-L1 = programmed death-ligand 1; CLDN18.2 = Claudin 18.2 Driving Accelerated Development and Value Creation of Innovations in a Quality-oriented and Cross-therapeutic Approach Our Platform-based Business Model Accelerate innovative asset development with at scale value creation Emerging innovation for asset sourcing Attractive �exit/spin-off Deeply rooted in emerging innovation ecosystem for long-time, with incomparable understanding of team capabilities and quality of assets from local innovators Scientific Translation Clinical De-risking NovaBridge Platform Oncology Co Ophthal Co Future New Co Givastomig(CLDN18.2 x 4-1BB bsAb) Ragistomig(PD-L1 x 4-1BB bsAb) Uliledlimab(CD73 mAb) … VIS-101(VEGF x Ang-2 bsAb) … … Strong exit through acquisition or robust options of financing to fuel continued growth and value creation
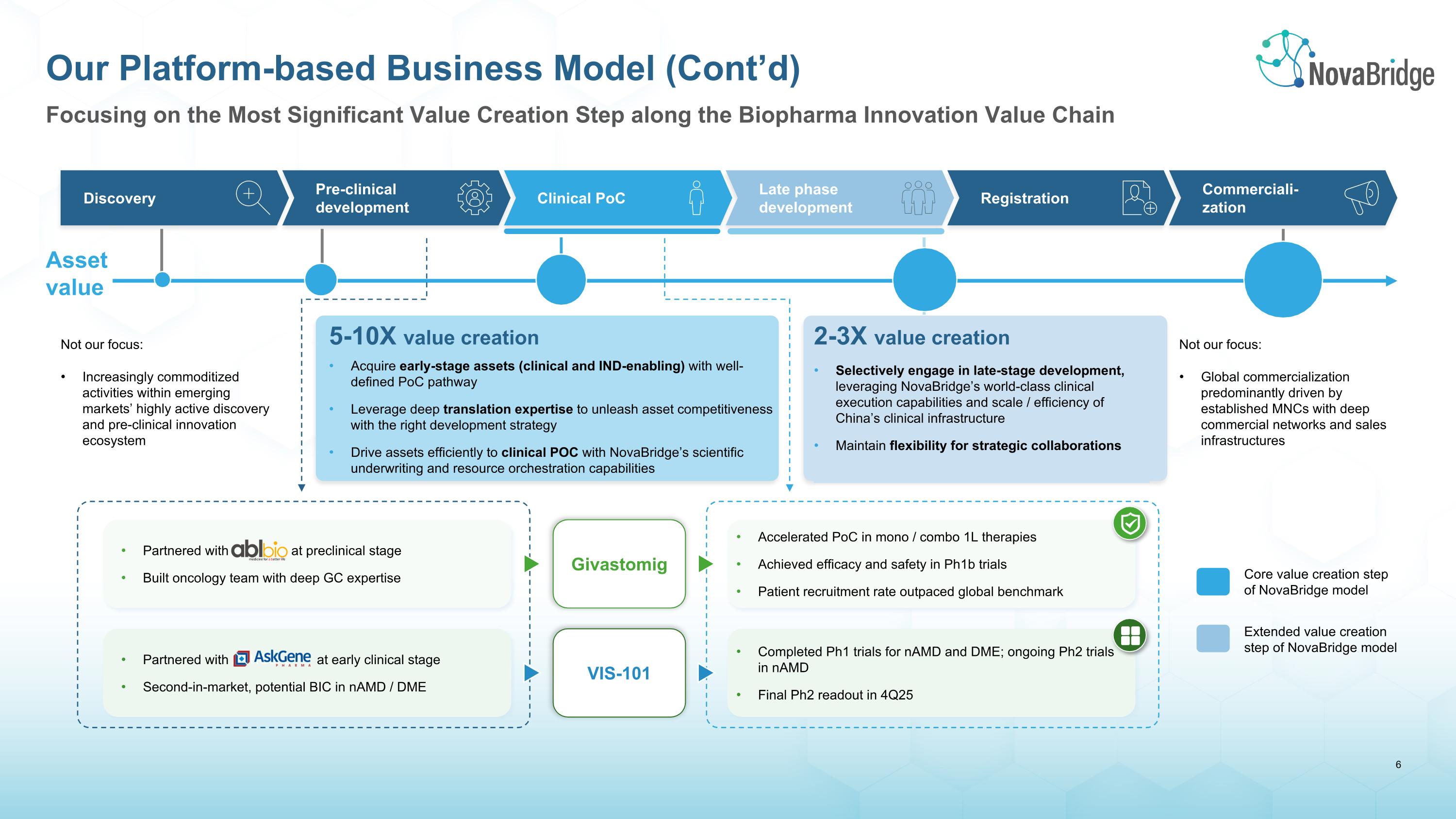
Focusing on the Most Significant Value Creation Step along the Biopharma Innovation Value Chain Our Platform-based Business Model (Cont’d) Discovery Pre-clinical �development Clinical PoC Late phase �development Registration Commerciali-�zation Not our focus: Increasingly commoditized activities within emerging markets’ highly active discovery and pre-clinical innovation ecosystem 5-10X value creation Acquire early-stage assets (clinical and IND-enabling) with well-defined PoC pathway Leverage deep translation expertise to unleash asset competitiveness with the right development strategy Drive assets efficiently to clinical POC with NovaBridge’s scientific underwriting and resource orchestration capabilities 2-3X value creation Selectively engage in late-stage development, leveraging NovaBridge’s world-class clinical execution capabilities and scale / efficiency of China’s clinical infrastructure Maintain flexibility for strategic collaborations Not our focus: Global commercialization predominantly driven by established MNCs with deep commercial networks and sales infrastructures Asset value Givastomig VIS-101 Accelerated PoC in mono / combo 1L therapies Achieved efficacy and safety in Ph1b trials Patient recruitment rate outpaced global benchmark Completed Ph1 trials for nAMD and DME; ongoing Ph2 trials in nAMD Final Ph2 readout in 4Q25 Partnered with ABL Bio at preclinical stage Built oncology team with deep GC expertise Partnered with ASKGene at early clinical stage Second-in-market, potential BIC in nAMD / DME Extended value creation �step of NovaBridge model Core value creation step �of NovaBridge model
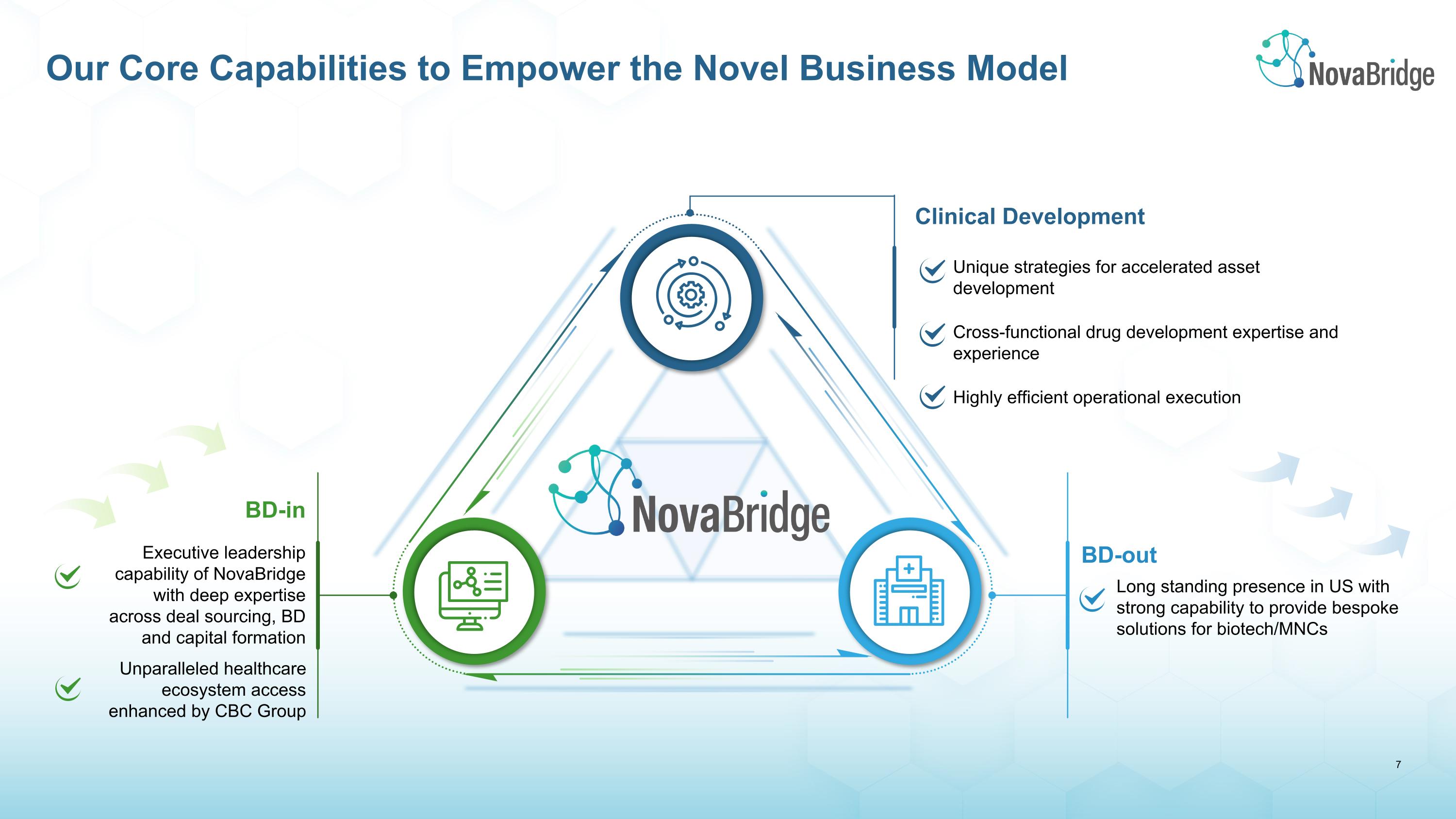
Our Core Capabilities to Empower the Novel Business Model BD-out Long standing presence in US with strong capability to provide bespoke solutions for biotech/MNCs Unparalleled healthcare ecosystem access enhanced by CBC Group Clinical Development Unique strategies for accelerated asset development Cross-functional drug development expertise and experience Highly efficient operational execution BD-in Executive leadership capability of NovaBridge with deep expertise across deal sourcing, BD and capital formation
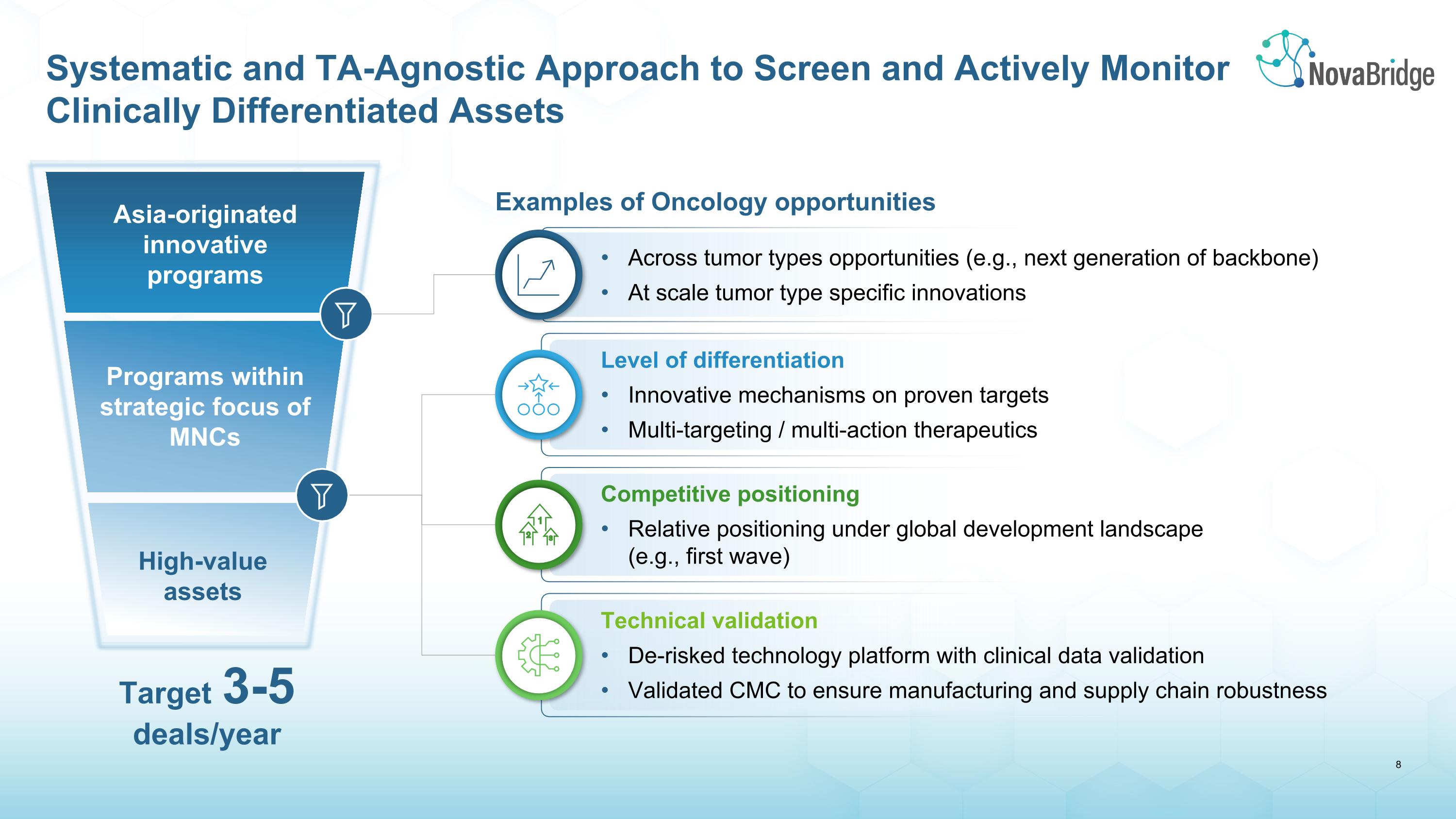
Systematic and TA-Agnostic Approach to Screen and Actively Monitor Clinically Differentiated Assets Target 3-5 �deals/year Asia-originated innovative programs High-value assets Programs within strategic focus of MNCs Examples of Oncology opportunities Across tumor types opportunities (e.g., next generation of backbone) At scale tumor type specific innovations Level of differentiation Innovative mechanisms on proven targets Multi-targeting / multi-action therapeutics Competitive positioning Relative positioning under global development landscape �(e.g., first wave) Technical validation De-risked technology platform with clinical data validation Validated CMC to ensure manufacturing and supply chain robustness
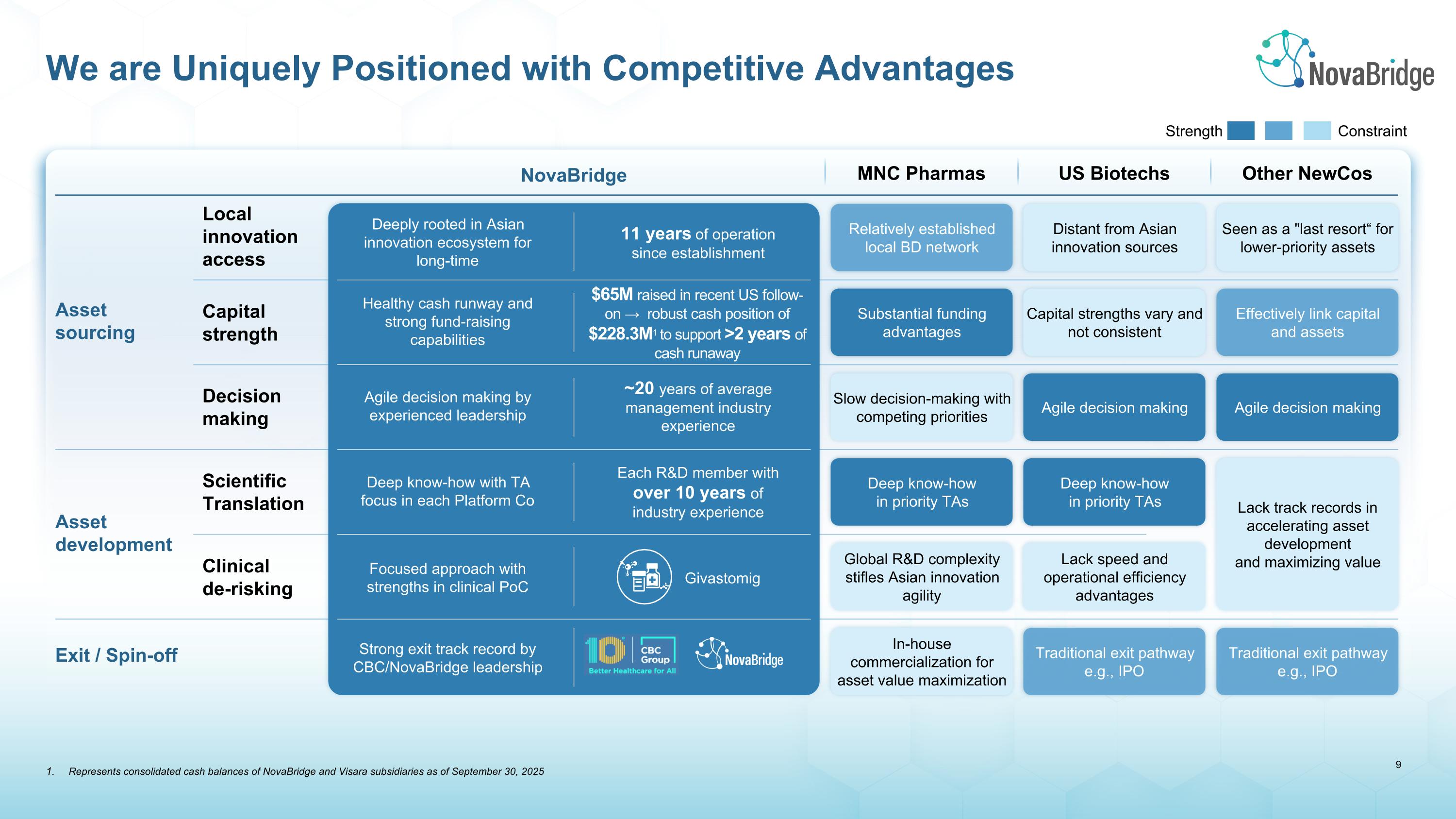
We are Uniquely Positioned with Competitive Advantages Strength Constraint Asset sourcing MNC Pharmas Clinical de-risking Scientific Translation US Biotechs Capital strength Decision making Local innovation access Asset development Other NewCos NovaBridge Exit / Spin-off Deep know-how in priority TAs Deep know-how in priority TAs Relatively established local BD network Distant from Asian innovation sources Lack track records in accelerating asset development and maximizing value Seen as a "last resort“ for lower-priority assets Substantial funding advantages Capital strengths vary and not consistent Effectively link capital and assets Slow decision-making with competing priorities Agile decision making Agile decision making Global R&D complexity stifles Asian innovation agility Lack speed and operational efficiency advantages In-house commercialization for asset value maximization Traditional exit pathway e.g., IPO Traditional exit pathway e.g., IPO Deep know-how with TA focus in each Platform Co Deeply rooted in Asian innovation ecosystem for long-time Healthy cash runway and strong fund-raising capabilities Agile decision making by experienced leadership Focused approach with strengths in clinical PoC Strong exit track record by CBC/NovaBridge leadership Each R&D member with over 10 years of industry experience 11 years of operation since establishment ~20 years of average management industry experience Givastomig $65M raised in recent US follow-on → robust cash position of $228.3M1 to support >2 years of cash runaway Represents consolidated cash balances of NovaBridge and Visara subsidiaries as of September 30, 2025
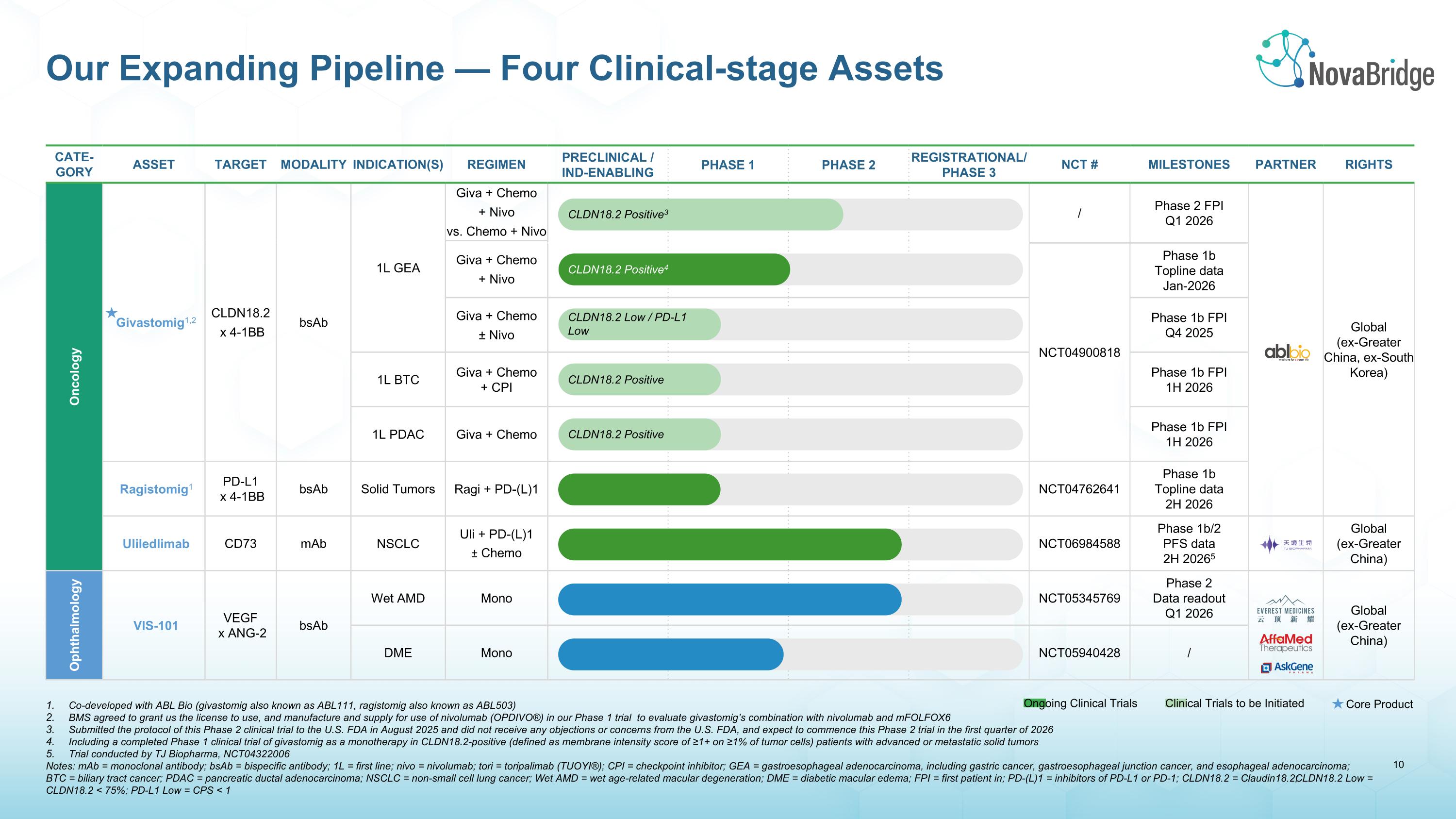
Co-developed with ABL Bio (givastomig also known as ABL111, ragistomig also known as ABL503) BMS agreed to grant us the license to use, and manufacture and supply for use of nivolumab (OPDIVO®) in our Phase 1 trial to evaluate givastomig’s combination with nivolumab and mFOLFOX6 Submitted the protocol of this Phase 2 clinical trial to the U.S. FDA in August 2025 and did not receive any objections or concerns from the U.S. FDA, and expect to commence this Phase 2 trial in the first quarter of 2026 Including a completed Phase 1 clinical trial of givastomig as a monotherapy in CLDN18.2-positive (defined as membrane intensity score of ≥1+ on ≥1% of tumor cells) patients with advanced or metastatic solid tumors Trial conducted by TJ Biopharma, NCT04322006 Notes: mAb = monoclonal antibody; bsAb = bispecific antibody; 1L = first line; nivo = nivolumab; tori = toripalimab (TUOYI®); CPI = checkpoint inhibitor; GEA = gastroesophageal adenocarcinoma, including gastric cancer, gastroesophageal junction cancer, and esophageal adenocarcinoma; �BTC = biliary tract cancer; PDAC = pancreatic ductal adenocarcinoma; NSCLC = non-small cell lung cancer; Wet AMD = wet age-related macular degeneration; DME = diabetic macular edema; FPI = first patient in; PD-(L)1 = inhibitors of PD-L1 or PD-1; CLDN18.2 = Claudin18.2; CLDN18.2 Low = CLDN18.2 < 75%; PD-L1 Low = CPS < 1 Our Expanding Pipeline — Four Clinical-stage Assets Cate-gory Asset Target Modality Indication(s) Regimen PRECLINICAL / IND-ENABLING PHASE 1 PHASE 2 REGISTRATIONAL/PHASE 3 NCT # MILESTONES PARTNER RIGHTS Oncology Givastomig1,2 CLDN18.2 x 4-1BB bsAb 1L GEA Giva + Chemo + Nivo vs. Chemo + Nivo / Phase 2 FPI Q1 2026 Global (ex-Greater China, ex-South Korea) Giva + Chemo + Nivo NCT04900818 Phase 1b Topline data Jan-2026 Giva + Chemo ± Nivo Phase 1b FPI Q4 2025 1L BTC Giva + Chemo + CPI Phase 1b FPI 1H 2026 1L PDAC Giva + Chemo Phase 1b FPI 1H 2026 Ragistomig1 PD-L1 x 4-1BB bsAb Solid Tumors Ragi + PD-(L)1 NCT04762641 Phase 1b Topline data 2H 2026 Uliledlimab CD73 mAb NSCLC Uli + PD-(L)1 ± Chemo NCT06984588 Phase 1b/2 PFS data 2H 20265 Global (ex-Greater China) Ophthalmology VIS-101 VEGF x ANG-2 bsAb Wet AMD Mono NCT05345769 Phase 2 Data readout Q1 2026 Global (ex-Greater China) DME Mono NCT05940428 / CLDN18.2 Positive4 Core Product Ongoing Clinical Trials Clinical Trials to be Initiated CLDN18.2 Low / PD-L1 Low CLDN18.2 Positive3 CLDN18.2 Positive CLDN18.2 Positive

Visionary and Seasoned Management Team We assembled a seasoned management team composed of industry veterans with extensive regional and functional expertise Years of Industry Experience # 11 Wei Fu Director and Executive �Chairman of our Board 24 Sean Fu PhD, MBA Chief Executive Officer Phillip Dennis MD, PhD Chief Medical Officer 27 Kyler Lei Chief Financial Officer 7 Sean Cao PhD Chief Business Development Officer 28 Claire Xu MD, PhD Senior Vice President, Clinical Development 17

Oncology Program Givastomig Claudin 18.2 X 4-1BB bsAb with Best-in-Class Potential
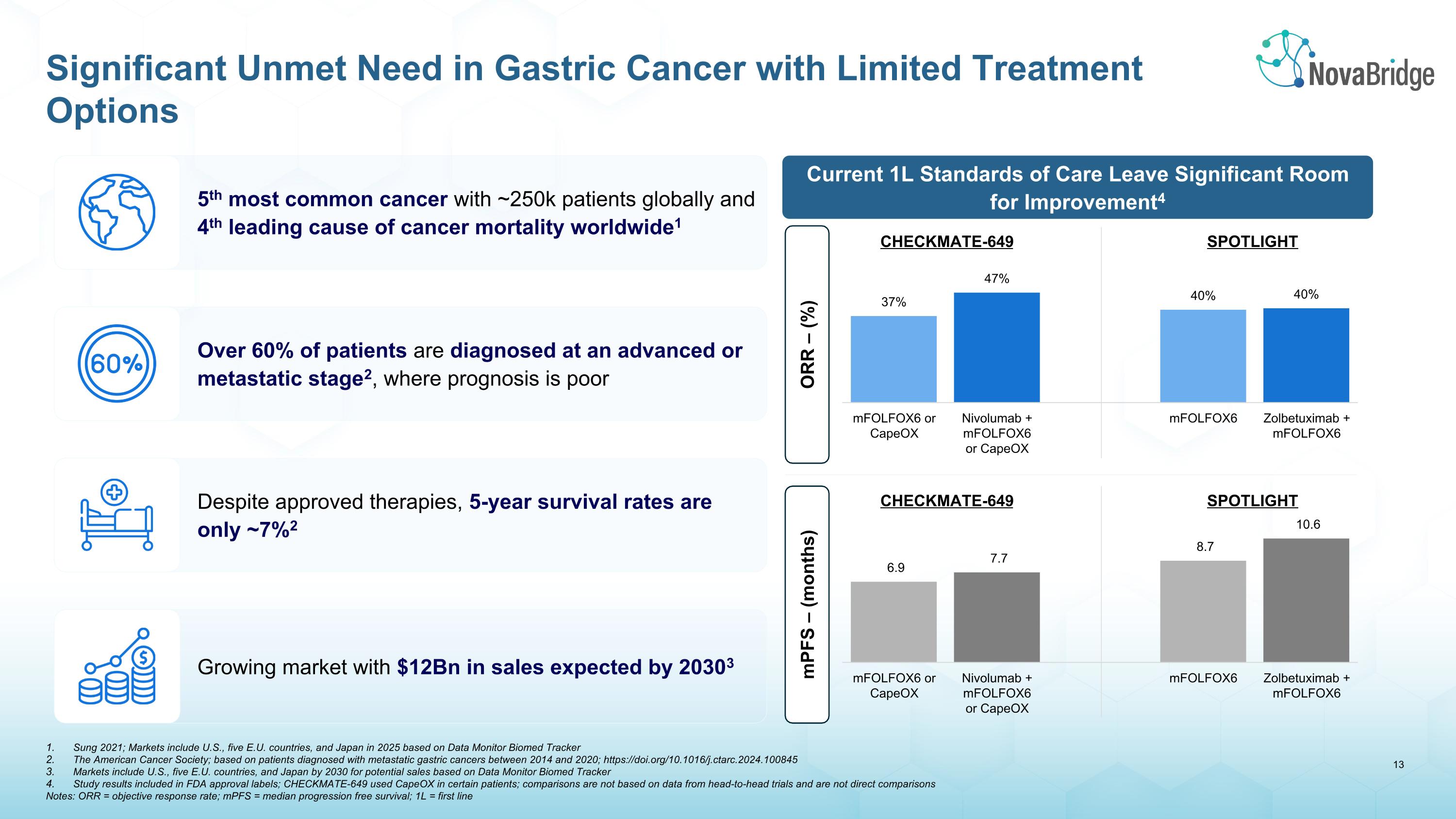
5th most common cancer with ~250k patients globally and 4th leading cause of cancer mortality worldwide1 Over 60% of patients are diagnosed at an advanced or metastatic stage2, where prognosis is poor Despite approved therapies, 5-year survival rates are only ~7%2 Growing market with $12Bn in sales expected by 20303 Sung 2021; Markets include U.S., five E.U. countries, and Japan in 2025 based on Data Monitor Biomed Tracker The American Cancer Society; based on patients diagnosed with metastatic gastric cancers between 2014 and 2020; https://doi.org/10.1016/j.ctarc.2024.100845 Markets include U.S., five E.U. countries, and Japan by 2030 for potential sales based on Data Monitor Biomed Tracker Study results included in FDA approval labels; CHECKMATE-649 used CapeOX in certain patients; comparisons are not based on data from head-to-head trials and are not direct comparisons Notes: ORR = objective response rate; mPFS = median progression free survival; 1L = first line Significant Unmet Need in Gastric Cancer with Limited Treatment Options Current 1L Standards of Care Leave Significant Room for Improvement4 ORR – (%) mPFS – (months) CHECKMATE-649 SPOTLIGHT CHECKMATE-649 SPOTLIGHT
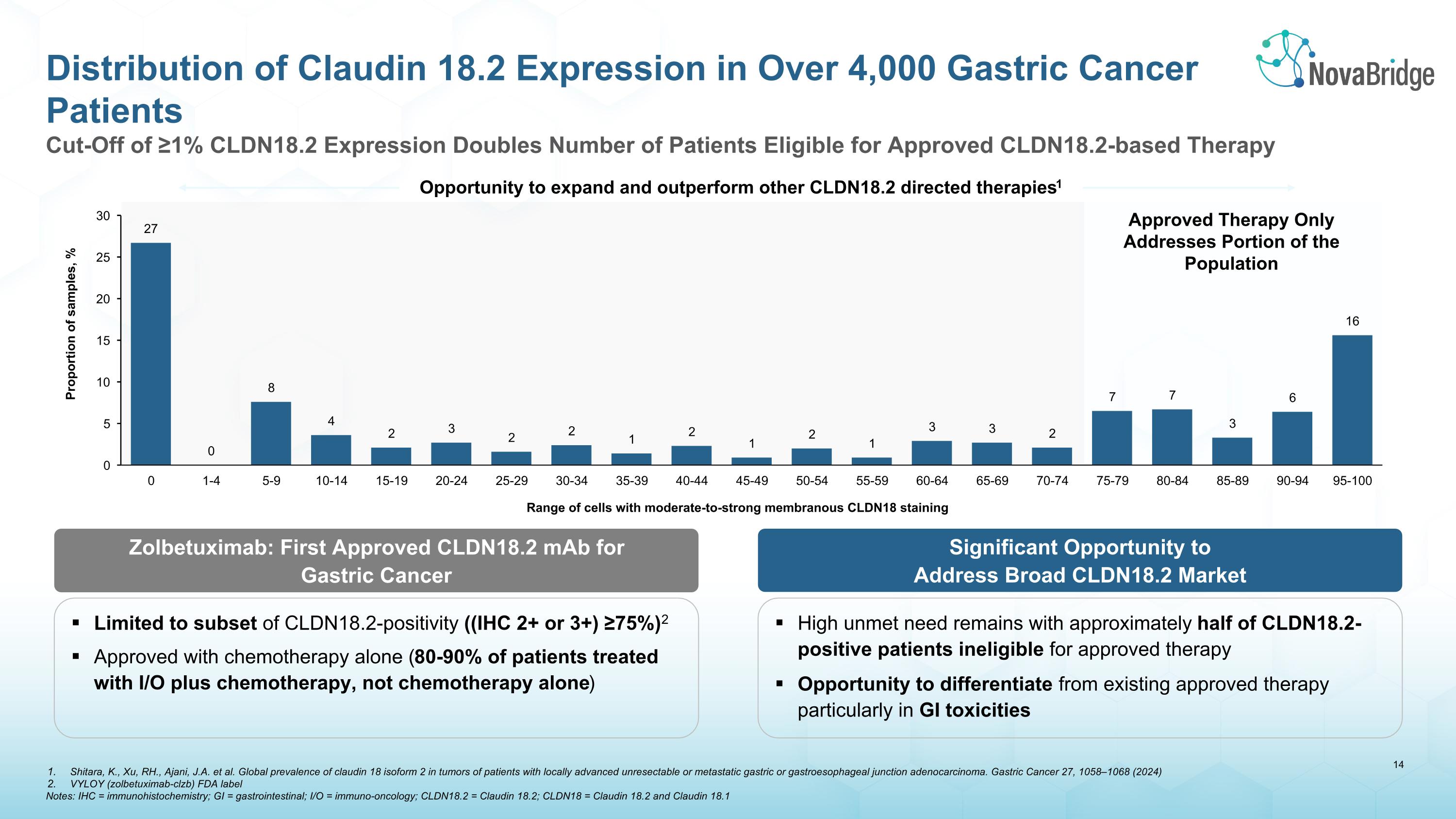
Zolbetuximab: First Approved CLDN18.2 mAb for Gastric Cancer Cut-Off of ≥1% CLDN18.2 Expression Doubles Number of Patients Eligible for Approved CLDN18.2-based Therapy Distribution of Claudin 18.2 Expression in Over 4,000 Gastric Cancer Patients Opportunity to expand and outperform other CLDN18.2 directed therapies1 Limited to subset of CLDN18.2-positivity ((IHC 2+ or 3+) ≥75%)2 Approved with chemotherapy alone (80-90% of patients treated with I/O plus chemotherapy, not chemotherapy alone) Significant Opportunity to Address Broad CLDN18.2 Market High unmet need remains with approximately half of CLDN18.2-positive patients ineligible for approved therapy Opportunity to differentiate from existing approved therapy particularly in GI toxicities Approved Therapy Only Addresses Portion of the Population Shitara, K., Xu, RH., Ajani, J.A. et al. Global prevalence of claudin 18 isoform 2 in tumors of patients with locally advanced unresectable or metastatic gastric or gastroesophageal junction adenocarcinoma. Gastric Cancer 27, 1058–1068 (2024) VYLOY (zolbetuximab-clzb) FDA label Notes: IHC = immunohistochemistry; GI = gastrointestinal; I/O = immuno-oncology; CLDN18.2 = Claudin 18.2; CLDN18 = Claudin 18.2 and Claudin 18.1

Revised from Nat Rev Clin Oncol. 2024 May;21(5):354-369 Notes: scFv = single chain Fragment-variable region; IgG1 = Immunoglobin G1; ADCC = antibody-dependent cell-mediated cytotoxicity; CDC = complement-dependent cytotoxicity; TME = tumor microenvironment; PFS = progression free survival; CLDN18.2 = Claudin 18.2; 1L = first line; mAb = monoclonal antibody;, PD-L1 = programmed death-ligand Givastomig: Broad Potential to Improve Standard-of-Care for Gastric Cancer and Other Solid Tumors Unique Molecular Design Balances Anti-Tumor Efficacy and Safety CLDN18.2 4-1BB scFv Robust ORR and promising emerging PFS data observed Activity in all patients, including those with low PD-L1 and/or low CLDN18.2 expression Well positioned with best-in-class potential to capture significant market opportunity in gastric cancer and beyond Favorable safety profile underpins the �1L line potential and broad therapeutic window Highly Potent CLDN18.2 mAb Silenced FC: IgG1 (N297A) Conditional 4-1 BB agonist Higher affinity than zolbetuximab Binds to tumor cells with a wide range of CLDN18.2 expression No ADCC or CDC Designed to minimize unintended systemic immune activation driven by FcgR-mediated 4-1BB clustering Localized T cell activation in TME leading to tumor killing and minimal 4-1BB-mediated liver toxicity or systemic immune response
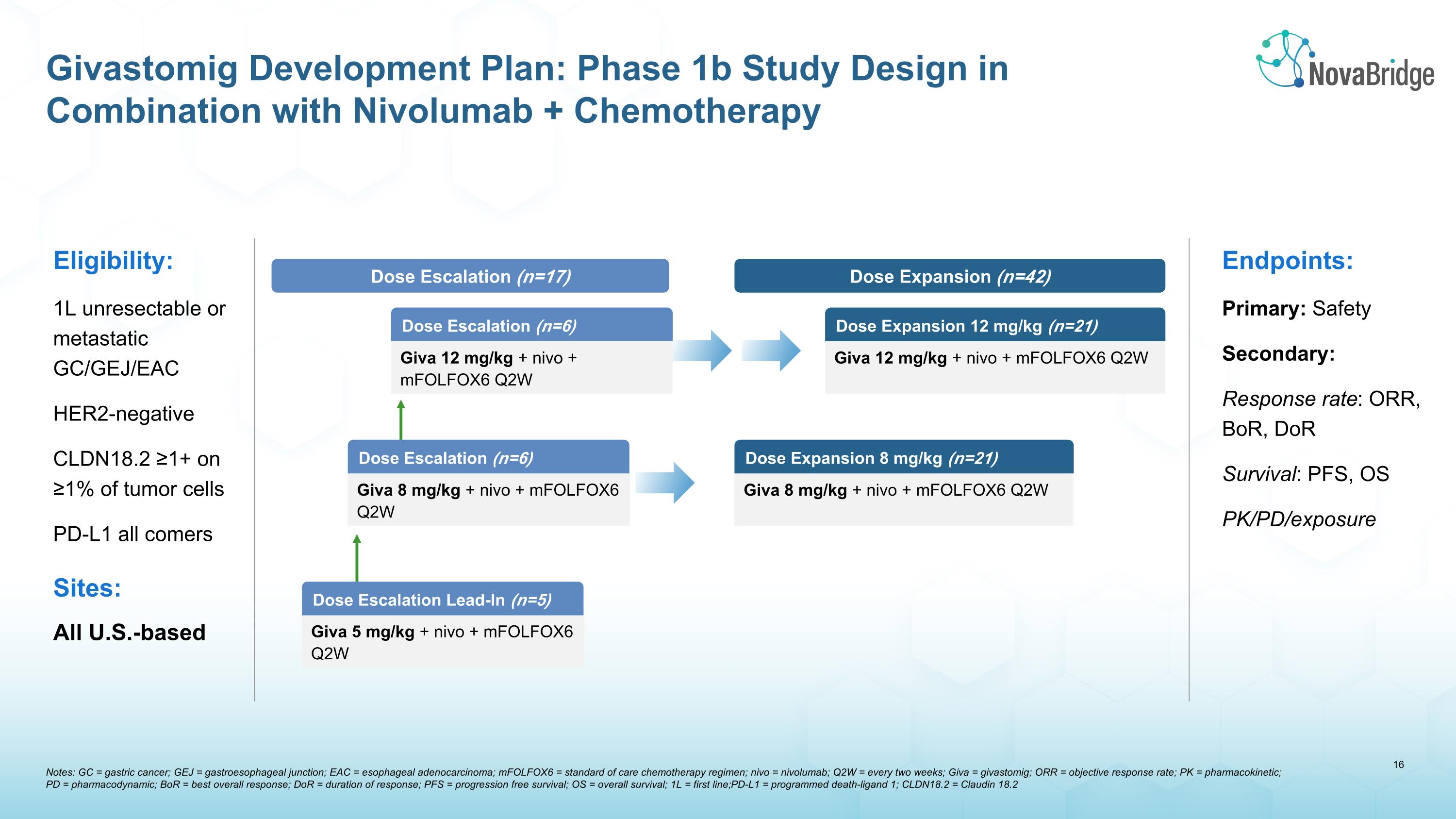
Notes: GC = gastric cancer; GEJ = gastroesophageal junction; EAC = esophageal adenocarcinoma; mFOLFOX6 = standard of care chemotherapy regimen; nivo = nivolumab; Q2W = every two weeks; Giva = givastomig; ORR = objective response rate; PK = pharmacokinetic; �PD = pharmacodynamic; BoR = best overall response; DoR = duration of response; PFS = progression free survival; OS = overall survival; 1L = first line; PD-L1 = programmed death-ligand 1; CLDN18.2 = Claudin 18.2 Givastomig Development Plan: Phase 1b Study Design in Combination with Nivolumab + Chemotherapy Eligibility: 1L unresectable or metastatic GC/GEJ/EAC HER2-negative CLDN18.2 ≥1+ on ≥1% of tumor cells PD-L1 all comers Sites: All U.S.-based Endpoints: Primary: Safety Secondary: Response rate: ORR, BoR, DoR Survival: PFS, OS PK/PD/exposure Dose Escalation Lead-In (n=5) Dose Escalation (n=17) Dose Expansion (n=42) Dose Escalation (n=6) Giva 12 mg/kg + nivo + mFOLFOX6 Q2W Dose Escalation (n=6) Giva 8 mg/kg + nivo + mFOLFOX6 Q2W Giva 5 mg/kg + nivo + mFOLFOX6 Q2W Dose Expansion 8 mg/kg (n=21) Giva 8 mg/kg + nivo + mFOLFOX6 Q2W Dose Expansion 12 mg/kg (n=21) Giva 12 mg/kg + nivo + mFOLFOX6 Q2W
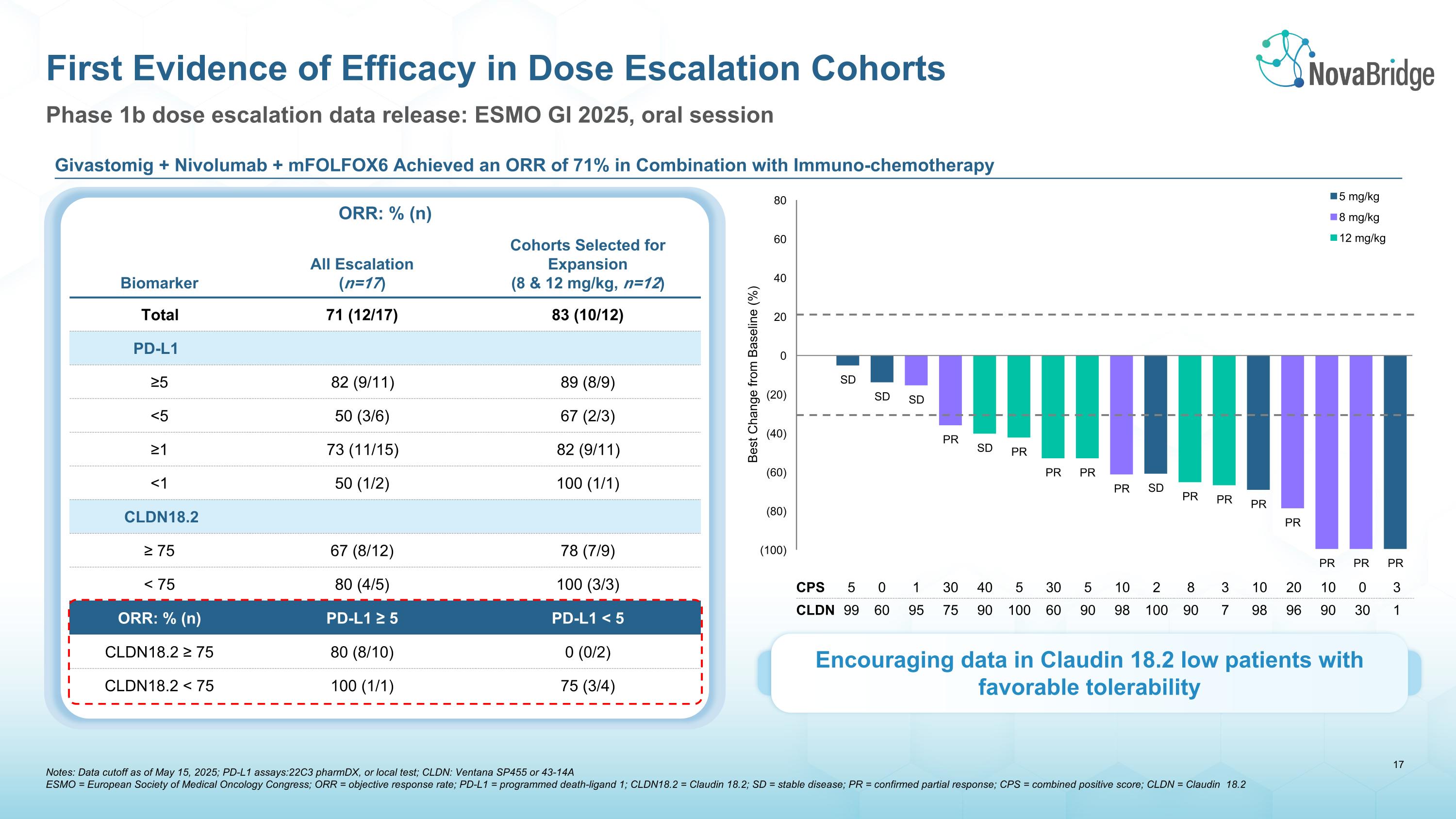
Phase 1b dose escalation data release: ESMO GI 2025, oral session First Evidence of Efficacy in Dose Escalation Cohorts ORR: % (n) Biomarker All Escalation�(n=17) Cohorts Selected for Expansion�(8 & 12 mg/kg, n=12) Total 71 (12/17) 83 (10/12) PD-L1 ≥5 82 (9/11) 89 (8/9) <5 50 (3/6) 67 (2/3) ≥1 73 (11/15) 82 (9/11) <1 50 (1/2) 100 (1/1) CLDN18.2 ≥ 75 67 (8/12) 78 (7/9) < 75 80 (4/5) 100 (3/3) ORR: % (n) PD-L1 ≥ 5 PD-L1 < 5 CLDN18.2 ≥ 75 80 (8/10) 0 (0/2) CLDN18.2 < 75 100 (1/1) 75 (3/4) CPS 5 0 1 30 40 5 30 5 10 2 8 3 10 20 10 0 3 CLDN 99 60 95 75 90 100 60 90 98 100 90 7 98 96 90 30 1 Givastomig + Nivolumab + mFOLFOX6 Achieved an ORR of 71% in Combination with Immuno-chemotherapy Encouraging data in Claudin 18.2 low patients with favorable tolerability Notes: Data cutoff as of May 15, 2025; PD-L1 assays:22C3 pharmDX, or local test; CLDN: Ventana SP455 or 43-14A ESMO = European Society of Medical Oncology Congress; ORR = objective response rate; PD-L1 = programmed death-ligand 1; CLDN18.2 = Claudin 18.2; SD = stable disease; PR = confirmed partial response; CPS = combined positive score; CLDN = Claudin 18.2
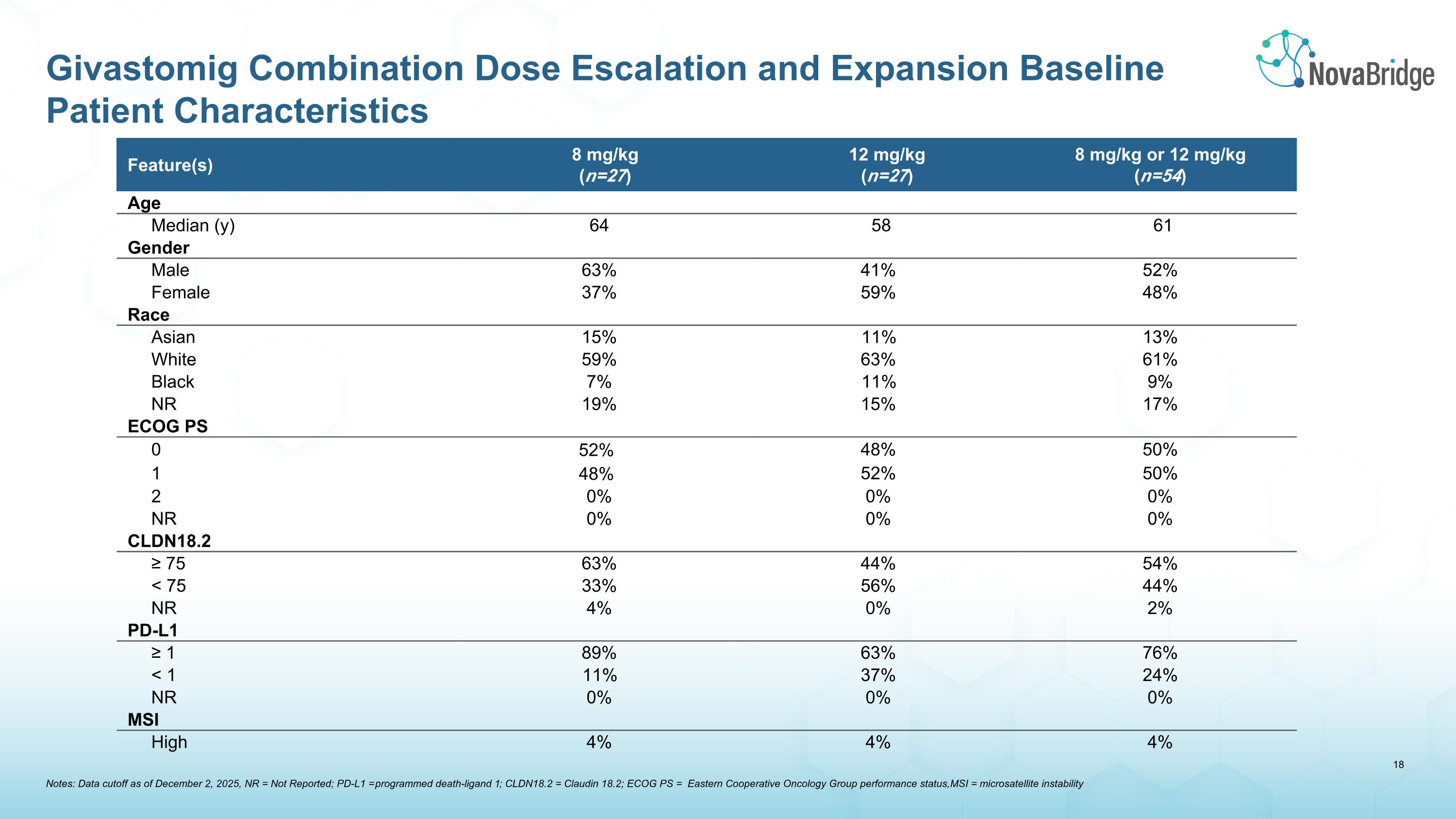
Feature(s) 8 mg/kg (n=27) Giva combo 12mg/kg (n=27) 12 mg/kg (n=27) 8 mg/kg or 12 mg/kg (n=54) Age Median (y) 64 58 61 Gender Male 63% 41% 52% Female 37% 59% 48% Race Asian 15% 11% 13% White 59% 63% 61% Black 7% 11% 9% NR 19% 15% 17% ECOG PS 0 52% 48% 50% 1 48% 52% 50% 2 0% 0% 0% NR 0% 0% 0% CLDN18.2 ≥ 75 63% 44% 54% < 75 33% 56% 44% NR 4% 0% 2% PD-L1 ≥ 1 89% 63% 76% < 1 11% 37% 24% NR 0% 0% 0% MSI High 4% 4% 4% Notes: Data cutoff as of December 2, 2025, NR = Not Reported; PD-L1 = programmed death-ligand 1; CLDN18.2 = Claudin 18.2; ECOG PS = Eastern Cooperative Oncology Group performance status, MSI = microsatellite instability Givastomig Combination Dose Escalation and Expansion Baseline Patient Characteristics
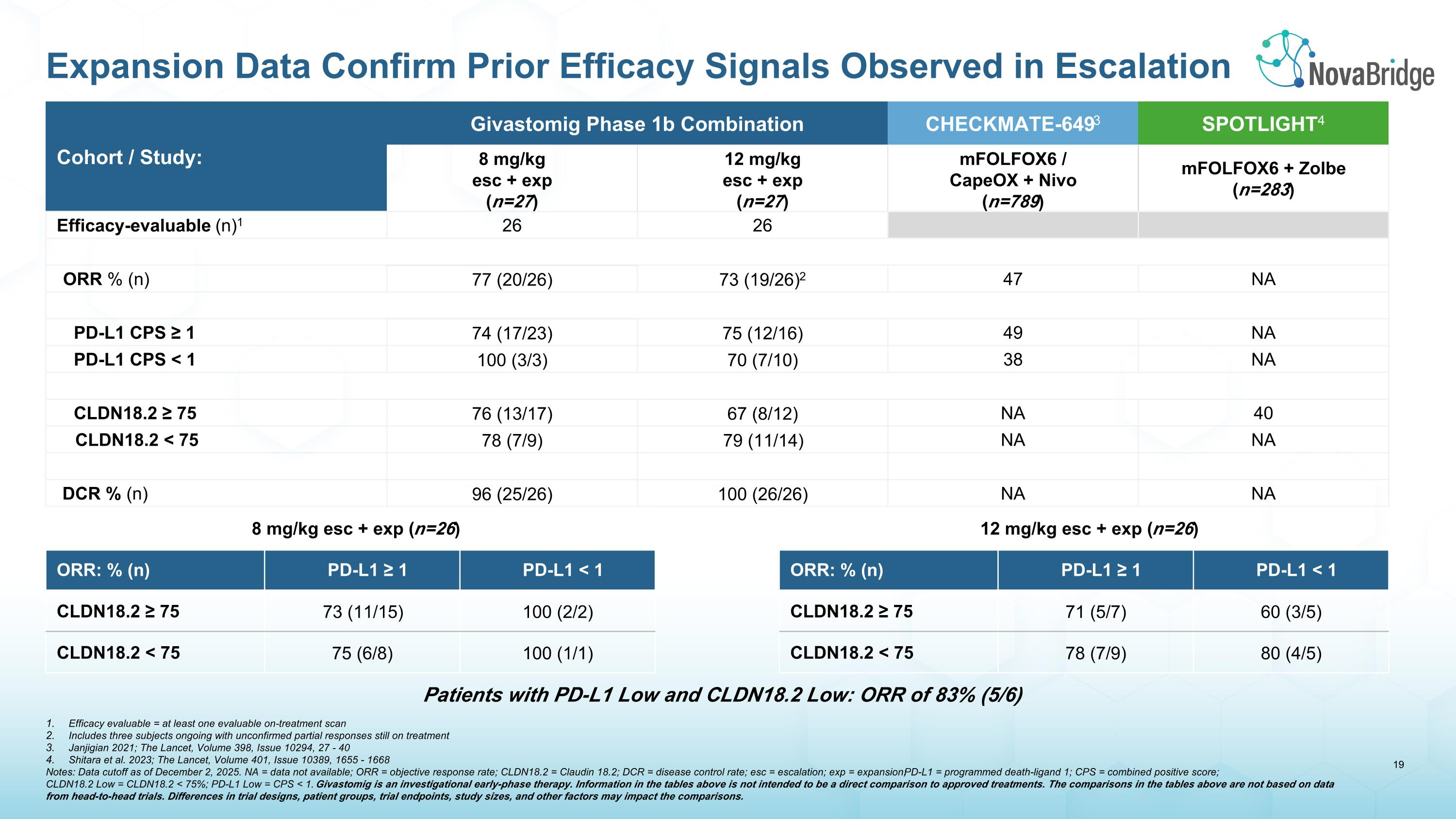
Cohort / Study: Givastomig Phase 1b Combination CHECKMATE-6493 SPOTLIGHT4 8 mg/kg esc + exp (n=27) 12 mg/kg esc + exp (n=27) mFOLFOX6 / �CapeOX + Nivo (n=789) mFOLFOX6 + Zolbe (n=283) Efficacy-evaluable (n)1 26 26 ORR % (n) 77 (20/26) 73 (19/26)2 47 NA PD-L1 CPS ≥ 1 74 (17/23) 75 (12/16) 49 NA PD-L1 CPS < 1 100 (3/3) 70 (7/10) 38 NA CLDN18.2 ≥ 75 76 (13/17) 67 (8/12) NA 40 CLDN18.2 < 75 78 (7/9) 79 (11/14) NA NA DCR % (n) 96 (25/26) 100 (26/26) NA NA 8 mg/kg esc + exp (n=26) ORR: % (n) PD-L1 ≥ 1 PD-L1 < 1 CLDN18.2 ≥ 75 73 (11/15) 100 (2/2) CLDN18.2 < 75 75 (6/8) 100 (1/1) 12 mg/kg esc + exp (n=26) ORR: % (n) PD-L1 ≥ 1 PD-L1 < 1 CLDN18.2 ≥ 75 71 (5/7) 60 (3/5) CLDN18.2 < 75 78 (7/9) 80 (4/5) Efficacy evaluable = at least one evaluable on-treatment scan Includes three subjects ongoing with unconfirmed partial responses still on treatment Janjigian 2021; The Lancet, Volume 398, Issue 10294, 27 - 40 Shitara et al. 2023; The Lancet, Volume 401, Issue 10389, 1655 - 1668 Notes: Data cutoff as of December 2, 2025. NA = data not available; ORR = objective response rate; CLDN18.2 = Claudin 18.2; DCR = disease control rate; esc = escalation; exp = expansion; PD-L1 = programmed death-ligand 1; CPS = combined positive score; �CLDN18.2 Low = CLDN18.2 < 75%; PD-L1 Low = CPS < 1. Givastomig is an investigational early-phase therapy. Information in the tables above is not intended to be a direct comparison to approved treatments. The comparisons in the tables above are not based on data from head-to-head trials. Differences in trial designs, patient groups, trial endpoints, study sizes, and other factors may impact the comparisons. Patients with PD-L1 Low and CLDN18.2 Low: ORR of 83% (5/6) Expansion Data Confirm Prior Efficacy Signals Observed in Escalation
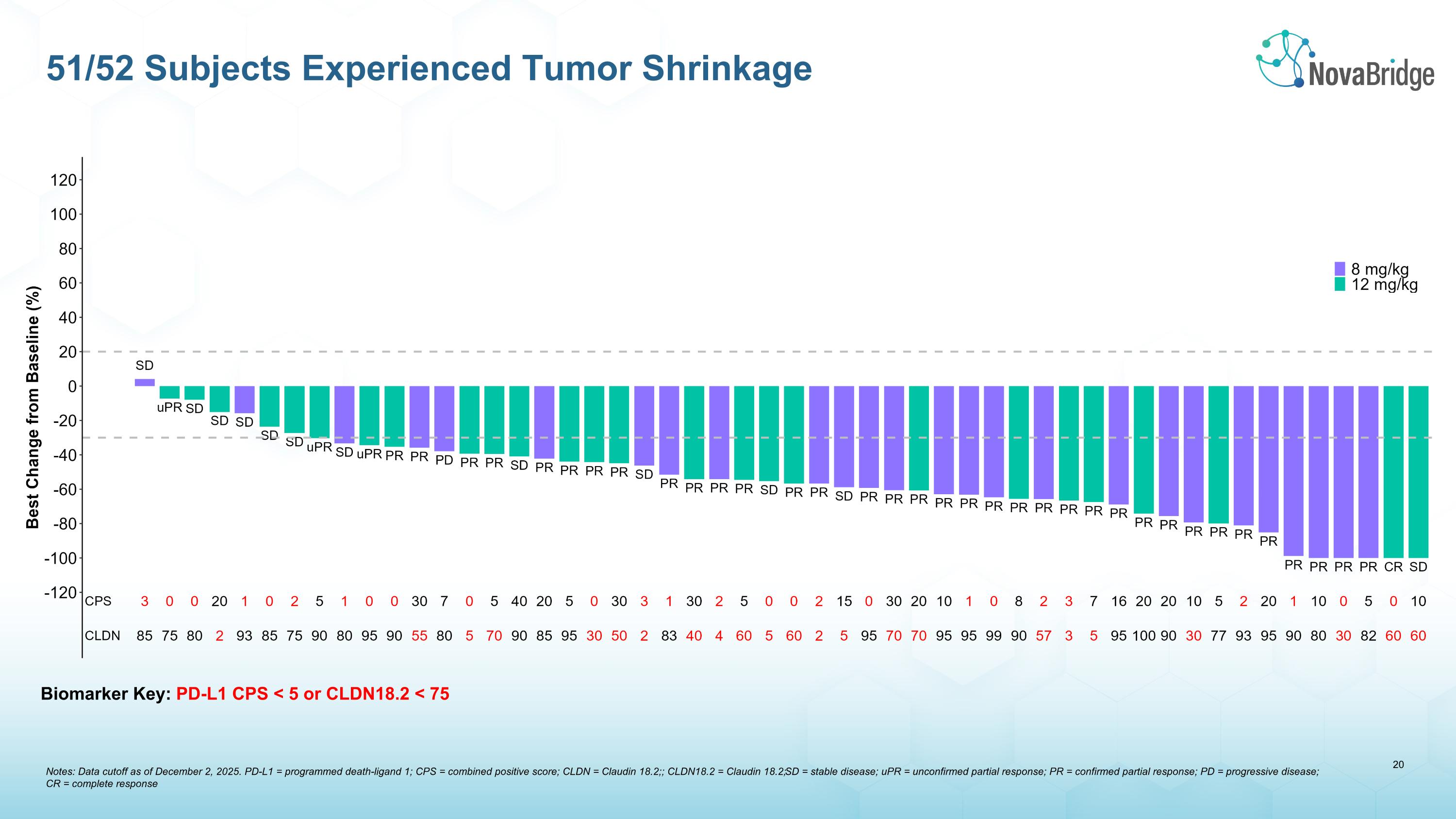
Biomarker Key: PD-L1 CPS < 5 or CLDN18.2 < 75 51/52 Subjects Experienced Tumor Shrinkage Notes: Data cutoff as of December 2, 2025. PD-L1 = programmed death-ligand 1; CPS = combined positive score; CLDN = Claudin 18.2;; CLDN18.2 = Claudin 18.2; SD = stable disease; uPR = unconfirmed partial response; PR = confirmed partial response; PD = progressive disease; �CR = complete response
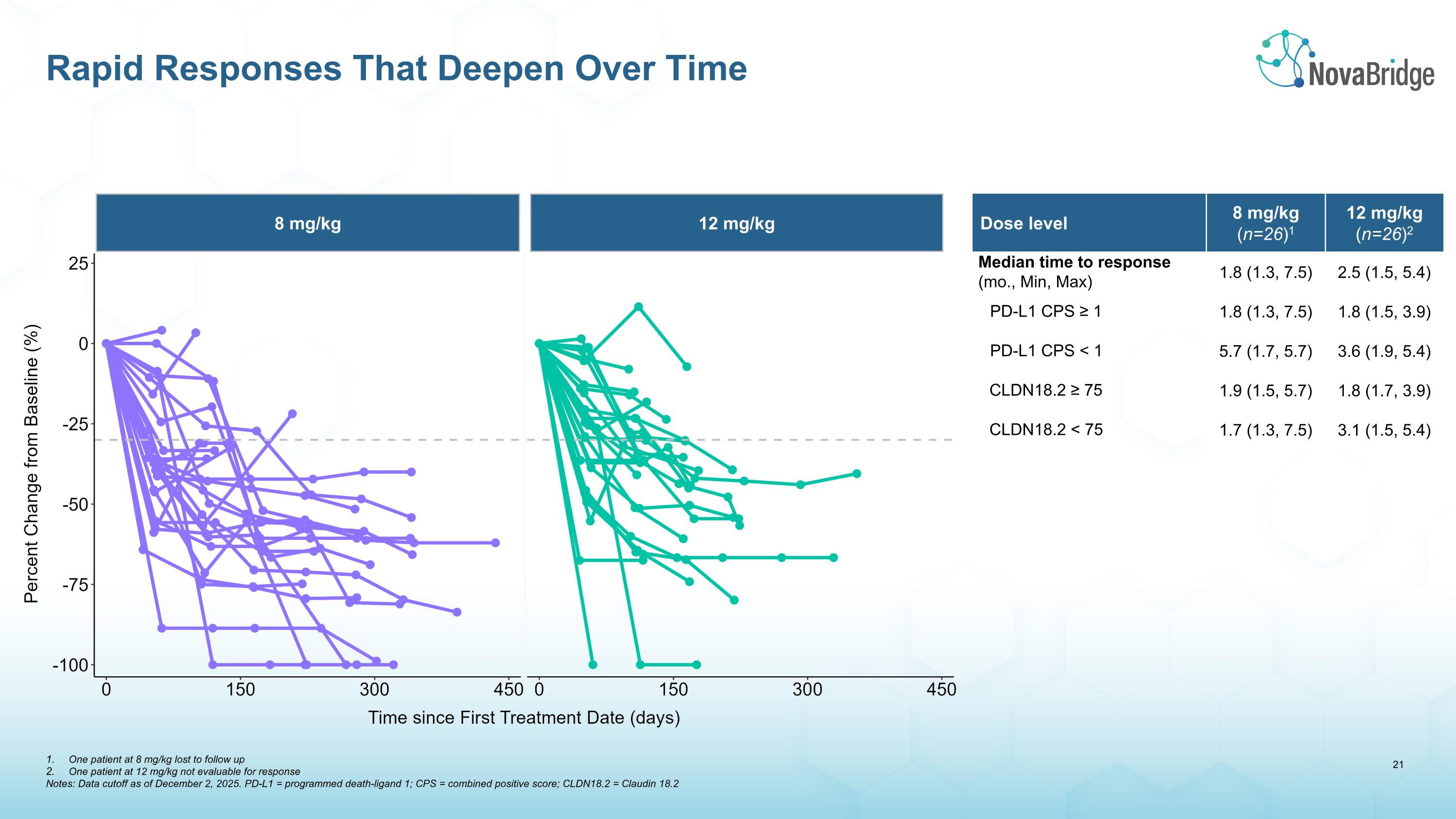
Rapid Responses That Deepen Over Time Dose level 8 mg/kg (n=26)1 12 mg/kg (n=26)2 Median time to response (mo., Min, Max) 1.8 (1.3, 7.5) 2.5 (1.5, 5.4) PD-L1 CPS ≥ 1 1.8 (1.3, 7.5) 1.8 (1.5, 3.9) PD-L1 CPS < 1 5.7 (1.7, 5.7) 3.6 (1.9, 5.4) CLDN18.2 ≥ 75 1.9 (1.5, 5.7) 1.8 (1.7, 3.9) CLDN18.2 < 75 1.7 (1.3, 7.5) 3.1 (1.5, 5.4) 8 mg/kg 12 mg/kg One patient at 8 mg/kg lost to follow up One patient at 12 mg/kg not evaluable for response Notes: Data cutoff as of December 2, 2025. PD-L1 = programmed death-ligand 1; CPS = combined positive score; CLDN18.2 = Claudin 18.2
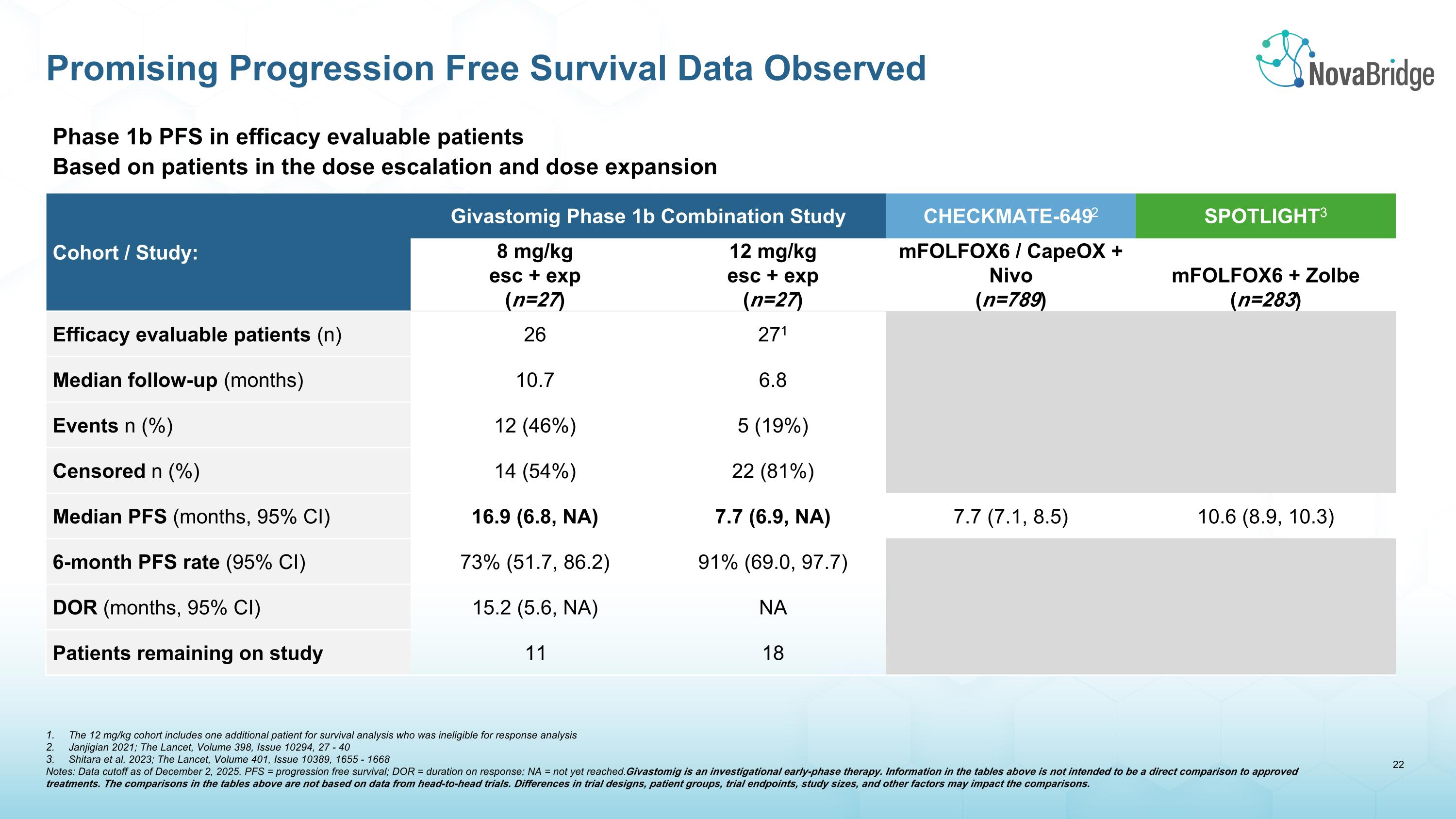
Phase 1b PFS in efficacy evaluable patients Based on patients in the dose escalation and dose expansion cohorts Cohort / Study: Givastomig Phase 1b Combination Study CHECKMATE-6492 SPOTLIGHT3 8 mg/kg esc + exp (n=27) 12 mg/kg esc + exp (n=27) mFOLFOX6 / CapeOX + Nivo (n=789) mFOLFOX6 + Zolbe (n=283) Efficacy evaluable patients (n) 26 271 Median follow-up (months) 10.7 6.8 Events n (%) 12 (46%) 5 (19%) Censored n (%) 14 (54%) 22 (81%) Median PFS (months, 95% CI) 16.9 (6.8, NA) 7.7 (6.9, NA) 7.7 (7.1, 8.5) 10.6 (8.9, 10.3) 6-month PFS rate (95% CI) 73% (51.7, 86.2) 91% (69.0, 97.7) DOR (months, 95% CI) 15.2 (5.6, NA) NA Patients remaining on study 11 18 Promising Progression Free Survival Data Observed The 12 mg/kg cohort includes one additional patient for survival analysis who was ineligible for response analysis Janjigian 2021; The Lancet, Volume 398, Issue 10294, 27 - 40 Shitara et al. 2023; The Lancet, Volume 401, Issue 10389, 1655 - 1668 Notes: Data cutoff as of December 2, 2025. PFS = progression free survival; DOR = duration on response; NA = not yet reached. Givastomig is an investigational early-phase therapy. Information in the tables above is not intended to be a direct comparison to approved treatments. The comparisons in the tables above are not based on data from head-to-head trials. Differences in trial designs, patient groups, trial endpoints, study sizes, and other factors may impact the comparisons.
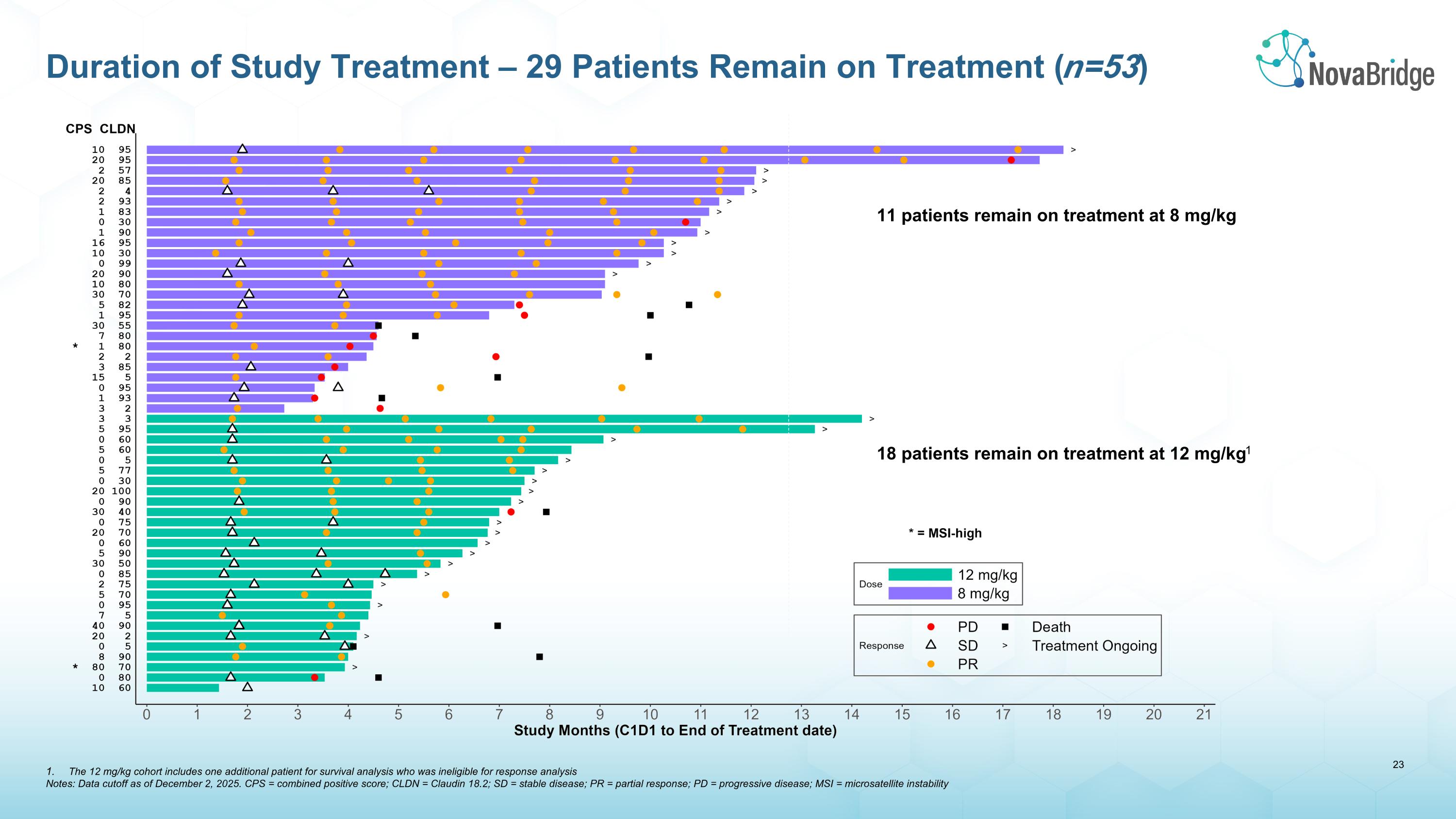
11 patients remain on treatment at 8 mg/kg 18 patients remain on treatment at 12 mg/kg1 Duration of Study Treatment – 29 Patients Remain on Treatment (n=53) The 12 mg/kg cohort includes one additional patient for survival analysis who was ineligible for response analysis Notes: Data cutoff as of December 2, 2025. CPS = combined positive score; CLDN = Claudin 18.2; SD = stable disease; PR = partial response; PD = progressive disease; MSI = microsatellite instability
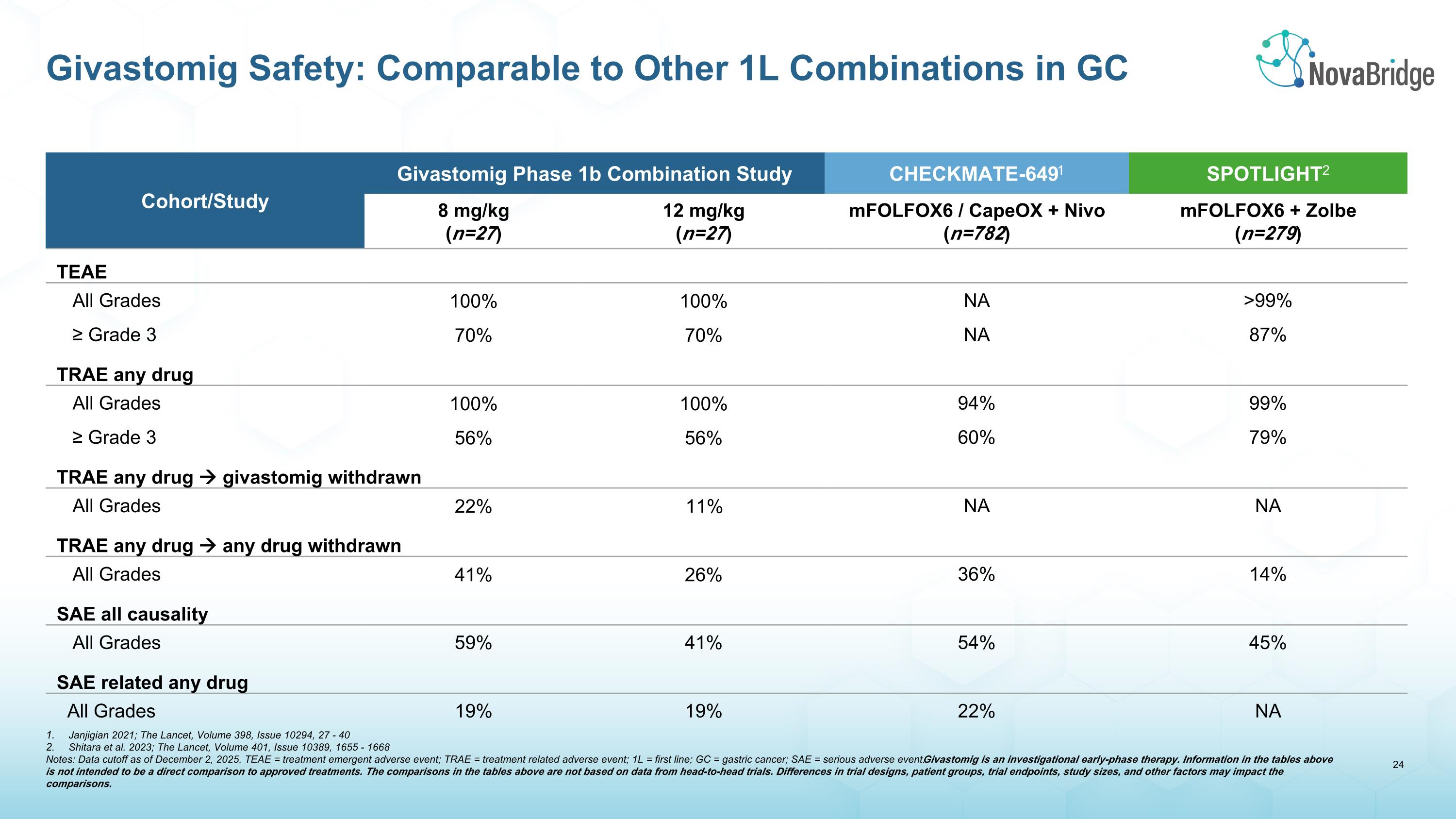
Cohort/Study Givastomig Phase 1b Combination Study CHECKMATE-6491 SPOTLIGHT2 8 mg/kg (n=27) 12 mg/kg (n=27) mFOLFOX6 / CapeOX + Nivo (n=782) mFOLFOX6 + Zolbe (n=279) TEAE All Grades 100% 100% NA >99% ≥ Grade 3 70% 70% NA 87% TRAE any drug All Grades 100% 100% 94% 99% ≥ Grade 3 56% 56% 60% 79% TRAE any drug givastomig withdrawn All Grades 22% 11% NA NA TRAE any drug any drug withdrawn All Grades 41% 26% 36% 14% SAE all causality All Grades 59% 41% 54% 45% SAE related any drug All Grades 19% 19% 22% NA Givastomig Safety: Comparable to Other 1L Combinations in GC Janjigian 2021; The Lancet, Volume 398, Issue 10294, 27 - 40 Shitara et al. 2023; The Lancet, Volume 401, Issue 10389, 1655 - 1668 Notes: Data cutoff as of December 2, 2025. TEAE = treatment emergent adverse event; TRAE = treatment related adverse event; 1L = first line; GC = gastric cancer; SAE = serious adverse event. Givastomig is an investigational early-phase therapy. Information in the tables above is not intended to be a direct comparison to approved treatments. The comparisons in the tables above are not based on data from head-to-head trials. Differences in trial designs, patient groups, trial endpoints, study sizes, and other factors may impact the comparisons.
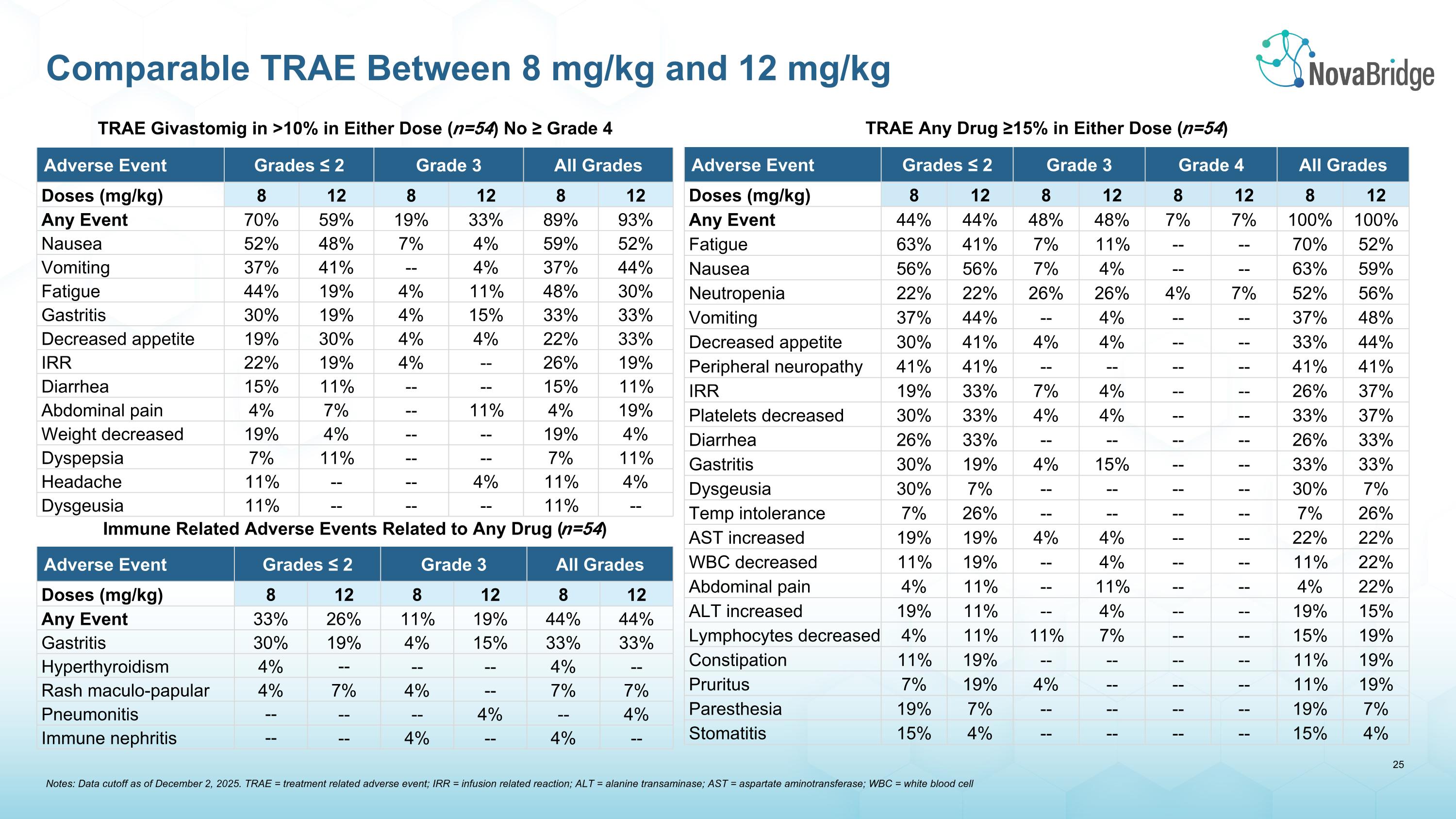
TRAE Any Drug ≥15% in Either Dose (n=54) Adverse Event Grades ≤ 2 Grade 3 Grade 4 All Grades Doses (mg/kg) 8 12 8 12 8 12 8 12 Any Event 44% 44% 48% 48% 7% 7% 100% 100% Fatigue 63% 41% 7% 11% -- -- 70% 52% Nausea 56% 56% 7% 4% -- -- 63% 59% Neutropenia 22% 22% 26% 26% 4% 7% 52% 56% Vomiting 37% 44% -- 4% -- -- 37% 48% Decreased appetite 30% 41% 4% 4% -- -- 33% 44% Peripheral neuropathy 41% 41% -- -- -- -- 41% 41% IRR 19% 33% 7% 4% -- -- 26% 37% Platelets decreased 30% 33% 4% 4% -- -- 33% 37% Diarrhea 26% 33% -- -- -- -- 26% 33% Gastritis 30% 19% 4% 15% -- -- 33% 33% Dysgeusia 30% 7% -- -- -- -- 30% 7% Temp intolerance 7% 26% -- -- -- -- 7% 26% AST increased 19% 19% 4% 4% -- -- 22% 22% WBC decreased 11% 19% -- 4% -- -- 11% 22% Abdominal pain 4% 11% -- 11% -- -- 4% 22% ALT increased 19% 11% -- 4% -- -- 19% 15% Lymphocytes decreased 4% 11% 11% 7% -- -- 15% 19% Constipation 11% 19% -- -- -- -- 11% 19% Pruritus 7% 19% 4% -- -- -- 11% 19% Paresthesia 19% 7% -- -- -- -- 19% 7% Stomatitis 15% 4% -- -- -- -- 15% 4% TRAE Givastomig in >10% in Either Dose (n=54) No ≥ Grade 4 Adverse Event Grades ≤ 2 Grade 3 All Grades Doses (mg/kg) 8 12 8 12 8 12 Any Event 70% 59% 19% 33% 89% 93% Nausea 52% 48% 7% 4% 59% 52% Vomiting 37% 41% -- 4% 37% 44% Fatigue 44% 19% 4% 11% 48% 30% Gastritis 30% 19% 4% 15% 33% 33% Decreased appetite 19% 30% 4% 4% 22% 33% IRR 22% 19% 4% -- 26% 19% Diarrhea 15% 11% -- -- 15% 11% Abdominal pain 4% 7% -- 11% 4% 19% Weight decreased 19% 4% -- -- 19% 4% Dyspepsia 7% 11% -- -- 7% 11% Headache 11% -- -- 4% 11% 4% Dysgeusia 11% -- -- -- 11% -- Immune Related Adverse Events Related to Any Drug (n=54) Adverse Event Grades ≤ 2 Grade 3 All Grades Doses (mg/kg) 8 12 8 12 8 12 Any Event 33% 26% 11% 19% 44% 44% Gastritis 30% 19% 4% 15% 33% 33% Hyperthyroidism 4% -- -- -- 4% -- Rash maculo-papular 4% 7% 4% -- 7% 7% Pneumonitis -- -- -- 4% -- 4% Immune nephritis -- -- 4% -- 4% -- Comparable TRAE Between 8 mg/kg and 12 mg/kg Notes: Data cutoff as of December 2, 2025. TRAE = treatment related adverse event; IRR = infusion related reaction; ALT = alanine transaminase; AST = aspartate aminotransferase; WBC = white blood cell
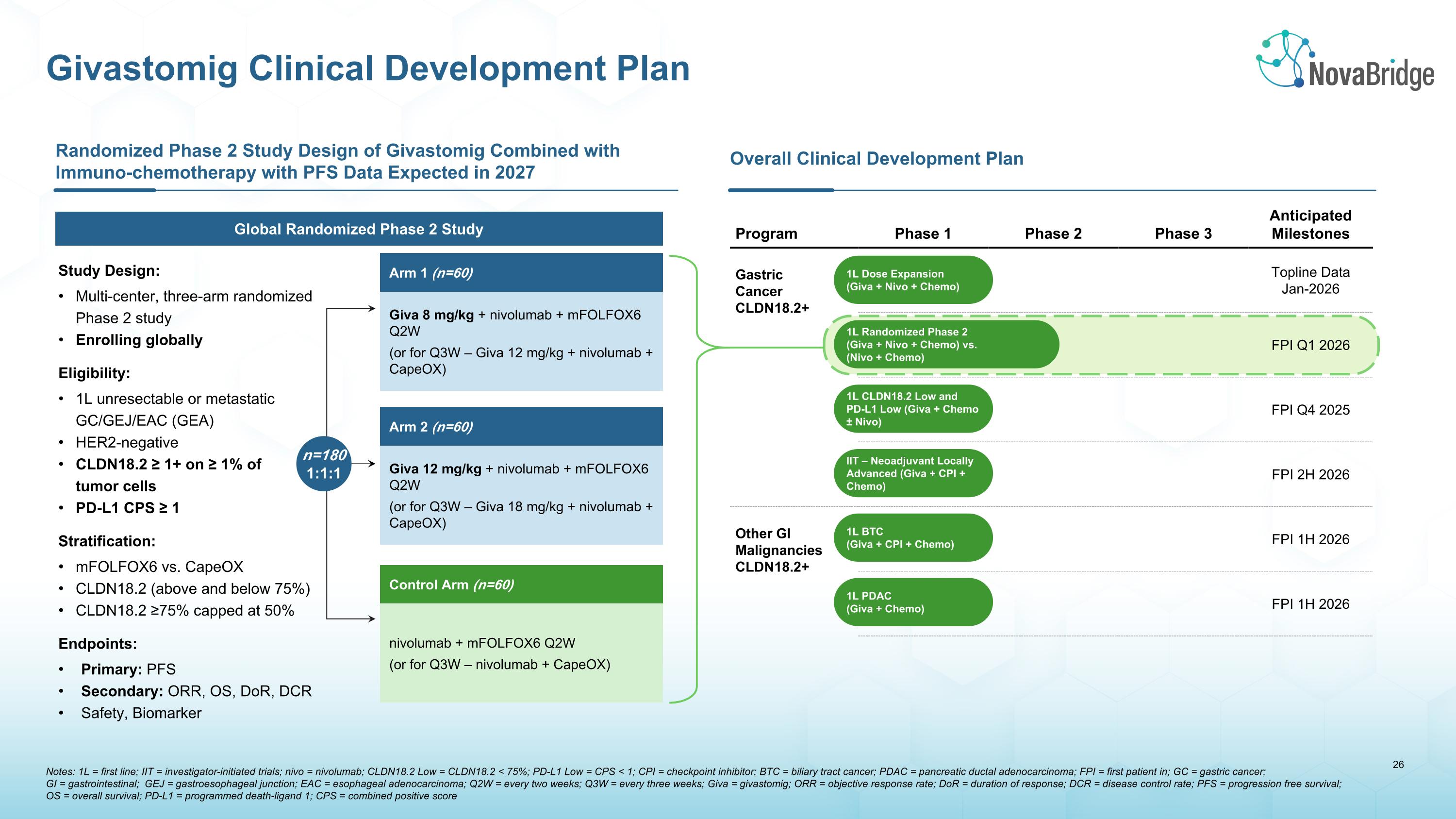
Overall Clinical Development Plan Program Phase 1 Phase 2 Phase 3 Anticipated�Milestones Gastric Cancer�CLDN18.2+ Topline Data�Jan-2026 FPI Q1 2026 FPI Q4 2025 FPI 2H 2026 Other GI�Malignancies CLDN18.2+ FPI 1H 2026 FPI 1H 2026 1L Dose Expansion (Giva + Nivo + Chemo) 1L Randomized Phase 2 (Giva + Nivo + Chemo) vs. �(Nivo + Chemo) 1L CLDN18.2 Low and PD-L1 Low (Giva + Chemo ± Nivo) IIT – Neoadjuvant Locally Advanced (Giva + CPI + Chemo) 1L BTC (Giva + CPI + Chemo) 1L PDAC (Giva + Chemo) Randomized Phase 2 Study Design of Givastomig Combined with Immuno-chemotherapy with PFS Data Expected in 2027 Study Design: Multi-center, three-arm randomized Phase 2 study Enrolling globally Eligibility: 1L unresectable or metastatic GC/GEJ/EAC (GEA) HER2-negative CLDN18.2 ≥ 1+ on ≥ 1% of �tumor cells PD-L1 CPS ≥ 1 Stratification: mFOLFOX6 vs. CapeOX CLDN18.2 (above and below 75%) CLDN18.2 ≥75% capped at 50% Endpoints: Primary: PFS Secondary: ORR, OS, DoR, DCR Safety, Biomarker Global Randomized Phase 2 Study Arm 1 (n=60) Giva 8 mg/kg + nivolumab + mFOLFOX6 Q2W (or for Q3W – Giva 12 mg/kg + nivolumab + CapeOX) Arm 2 (n=60) Giva 12 mg/kg + nivolumab + mFOLFOX6 Q2W (or for Q3W – Giva 18 mg/kg + nivolumab + CapeOX) Control Arm (n=60) nivolumab + mFOLFOX6 Q2W (or for Q3W – nivolumab + CapeOX) Notes: 1L = first line; IIT = investigator-initiated trials; nivo = nivolumab; CLDN18.2 Low = CLDN18.2 < 75%; PD-L1 Low = CPS < 1; CPI = checkpoint inhibitor; BTC = biliary tract cancer; PDAC = pancreatic ductal adenocarcinoma; FPI = first patient in; GC = gastric cancer; �GI = gastrointestinal; GEJ = gastroesophageal junction; EAC = esophageal adenocarcinoma; Q2W = every two weeks; Q3W = every three weeks; Giva = givastomig; ORR = objective response rate; DoR = duration of response; DCR = disease control rate; PFS = progression free survival; OS = overall survival; PD-L1 = programmed death-ligand 1; CPS = combined positive score Givastomig Clinical Development Plan n=180 1:1:1

Expansion Data Reinforces Givastomig's Best-in-Class Potential Robust efficacy, with 77% ORR observed at 8 mg/kg and 73% ORR observed at 12 mg/kg Responses observed across a wide range of PD-L1 and CLDN18.2 expression levels Durable responses with 16.9-month mPFS observed at 8 mg/kg; follow-up for 12 mg/kg is shorter and will be reported in 2026 Well tolerated in combination with nivolumab and mFOLFOX6, without dose dependent toxicity Broad potential in gastric cancer and other solid tumors Detailed Phase 1b expansion data expected to be presented at a medical conference in 2026 CLDN18.2 4-1BB scFv Notes: Data cutoff as of December 2, 2025. scFv = single chain Fragment-variable region; ORR = objective response rate; PD-L1 = programmed death-ligand 1; CLDN18.2 = Claudin 18.2; mPFS = median progression free survival
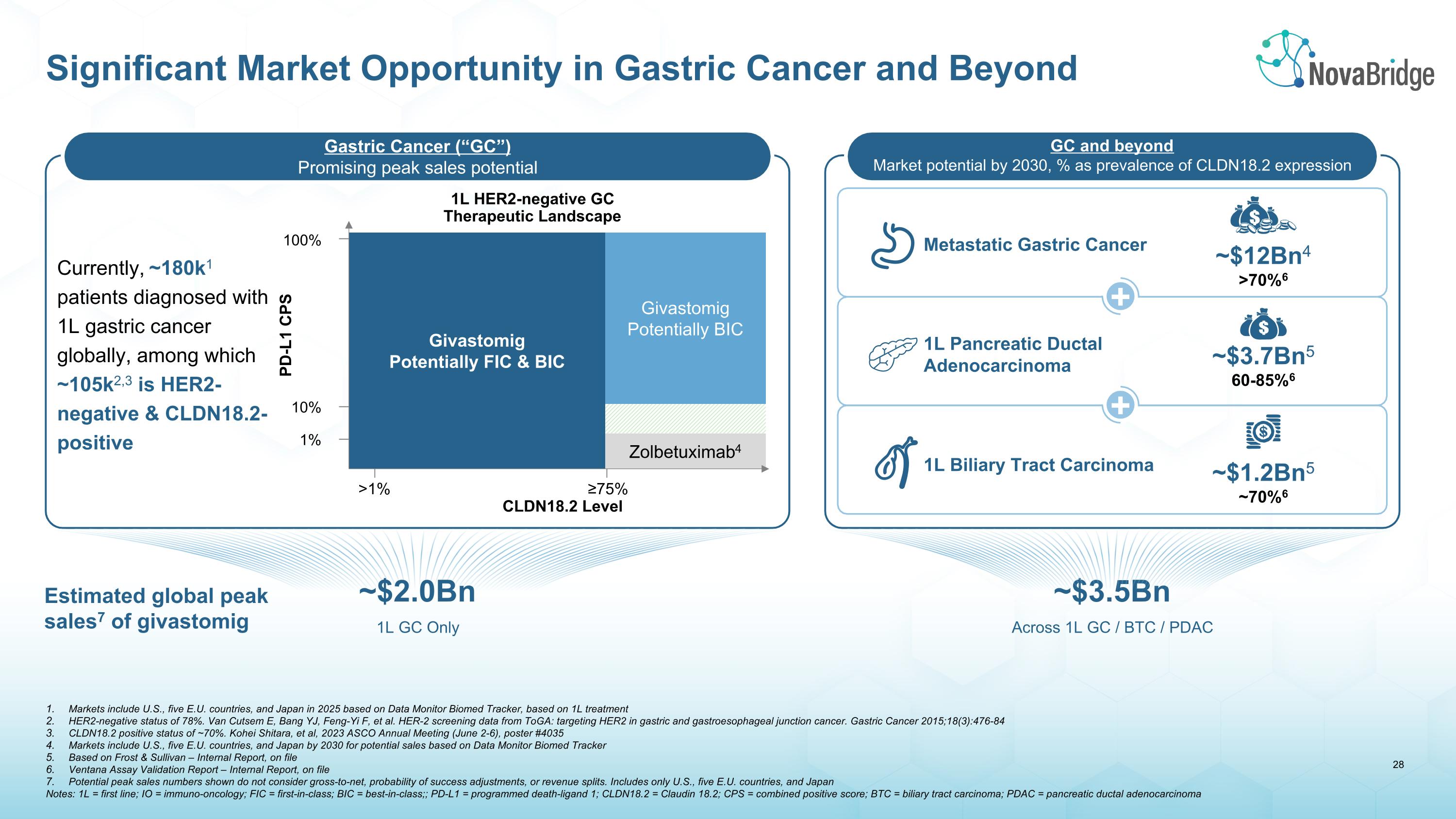
Markets include U.S., five E.U. countries, and Japan in 2025 based on Data Monitor Biomed Tracker, based on 1L treatment HER2-negative status of 78%. Van Cutsem E, Bang YJ, Feng-Yi F, et al. HER-2 screening data from ToGA: targeting HER2 in gastric and gastroesophageal junction cancer. Gastric Cancer 2015;18(3):476-84 CLDN18.2 positive status of ~70%. Kohei Shitara, et al, 2023 ASCO Annual Meeting (June 2-6), poster #4035 Markets include U.S., five E.U. countries, and Japan by 2030 for potential sales based on Data Monitor Biomed Tracker Based on Frost & Sullivan – Internal Report, on file Ventana Assay Validation Report – Internal Report, on file Potential peak sales numbers shown do not consider gross-to-net, probability of success adjustments, or revenue splits. Includes only U.S., five E.U. countries, and Japan Notes: 1L = first line; IO = immuno-oncology; FIC = first-in-class; BIC = best-in-class;; PD-L1 = programmed death-ligand 1; CLDN18.2 = Claudin 18.2; CPS = combined positive score; BTC = biliary tract carcinoma; PDAC = pancreatic ductal adenocarcinoma Significant Market Opportunity in Gastric Cancer and Beyond Givastomig�Potentially FIC & BIC Givastomig Potentially BIC Zolbetuximab4 100% 10% 1% >1% ≥75% CLDN18.2 Level PD-L1 CPS 1L HER2-negative GC�Therapeutic Landscape Metastatic Gastric Cancer ~$12Bn4 >70%6 1L Pancreatic Ductal Adenocarcinoma ~$3.7Bn5 60-85%6 1L Biliary Tract Carcinoma ~$1.2Bn5 ~70%6 ~$3.5Bn Across 1L GC / BTC / PDAC Gastric Cancer (“GC”) Promising peak sales potential GC and beyond Market potential by 2030, % as prevalence of CLDN18.2 expression + + Estimated global peak sales7 of givastomig ~$2.0Bn 1L GC Only Currently, ~180k1 patients diagnosed with 1L gastric cancer globally, among which ~105k2,3 is HER2-negative & CLDN18.2-positive

Ophthalmology Program VIS-101

Transaction Structure Visara, Inc. NovaBridge AffaMed ESOP 65% 30% 5% NovaBridge contributed cash in exchange for 65% of the equity interest in Visara AffaMed contributed its rights and interests in VIS-101 to Visara in exchange for 30% of the equity interest in Visara The remaining 5% equity interest in Visara reserved for an ESOP VIS-101-related ownership rights are shown above Emmett Cunningham MD, PhD, MPH Co-Founder and Executive Chairman World-renowned ophthalmologist; Former Senior Managing Director, Blackstone Group 25+ years of experience as an entrepreneur and investor Co-founder of 5+ companies, with a track record of serial entrepreneurial successes (IPO or acquired by MNCs) Internationally recognized specialist in infectious and inflammatory eye disease with over 450 publications Led the development of Macugen®: a first-in-class VEGF-A inhibitor for AMD and DME Cadmus Rich, CPE Chief Medical Officer Carlos Quezada-Ruiz MD, FASRS SAB Chairman 18+ years experience as an Executive level R&D professional with deep ophthalmology experience at multiple pharmaceutical and biotechnology companies including Lassen Therapeutics, Aura Biosciences and Alcon Strong experience working with FDA, EMA and MHRA on multiple, varied research and development projects NewCo Leadership Visara is Led by an Exceptional and Experienced Leadership Team 25+ years experience in ophthalmology holding various roles as a vitreoretinal surgeon, translational science & drug development executive, clinical R&D & TA head Most recently was the medical lead for VABYSMO® at Roche, and played a pivotal role in the global development and approvals of VABYSMO® and SUSIVMO ®, leading design, execution, readouts, fillings and launch

A novel VEGF × Ang-2 bsAb with the potential to emerge as a leading therapeutic option for multiple retinal indications that could lead to vision loss, including wet AMD, DME, and retinal vein occlusion VIS-101: Best-in-Class VEGF × Ang-2 Bispecific for Significant Ophthalmology Diseases with High Unmet Needs Current standard of care predominantly involves anti-VEGF therapies, significant unmet medical needs remain 30%-50% patients exhibit an inadequate response to available treatments Frequent injection requirements compromise long-term adherence, and patients may experience a reduction in therapeutic response over time or insufficient treatment durability Increases in durability have rapidly propelled revenue growth for successive generations of agents (ELYEA HD and VABYSMO) Phase 2a clinical findings suggest VIS-101 has the potential to become a more potent, second- and best-in-class VEGF × Ang-2 agent with superior durability A novel bsAb bioengineered with best-in-class potential by targeting VEGF × Ang-2 that features twice the binding sites of the benchmark product, delivering superior binding and blocking activity VIS-101 demonstrated superior VEGF × Ang-2 inhibitory activity compared to the benchmark product 2- and 6-fold increases in binding activity for VEGF and Ang-2 2- and 16-fold increases in inhibitory activity Increased revenue of anti-VEGF-A ophthalmology drugs correlated with superior pharmacokinetic inhibition and improved clinical durability LUCENTIS® Q4W EYLEA® Q8W VABYSMO® Q8-16W EYLEA HD® Q8-16W Revenue 2y from Launch $1.8Bn $2.8Bn $4.4Bn $1.5Bn VIS-101 Q20-24W (longer durability) Efficacy on pre-treated patients Next-Gen �SOC Less priming dose Anti-Ang-2 Anti-VEGF-A Modified Fc Source: Global data & Evaluate Pharma; sales revenue forecasts for LUCENTIS ®, EYLEA ®, VABYSMO ® and EYLEA HD ®
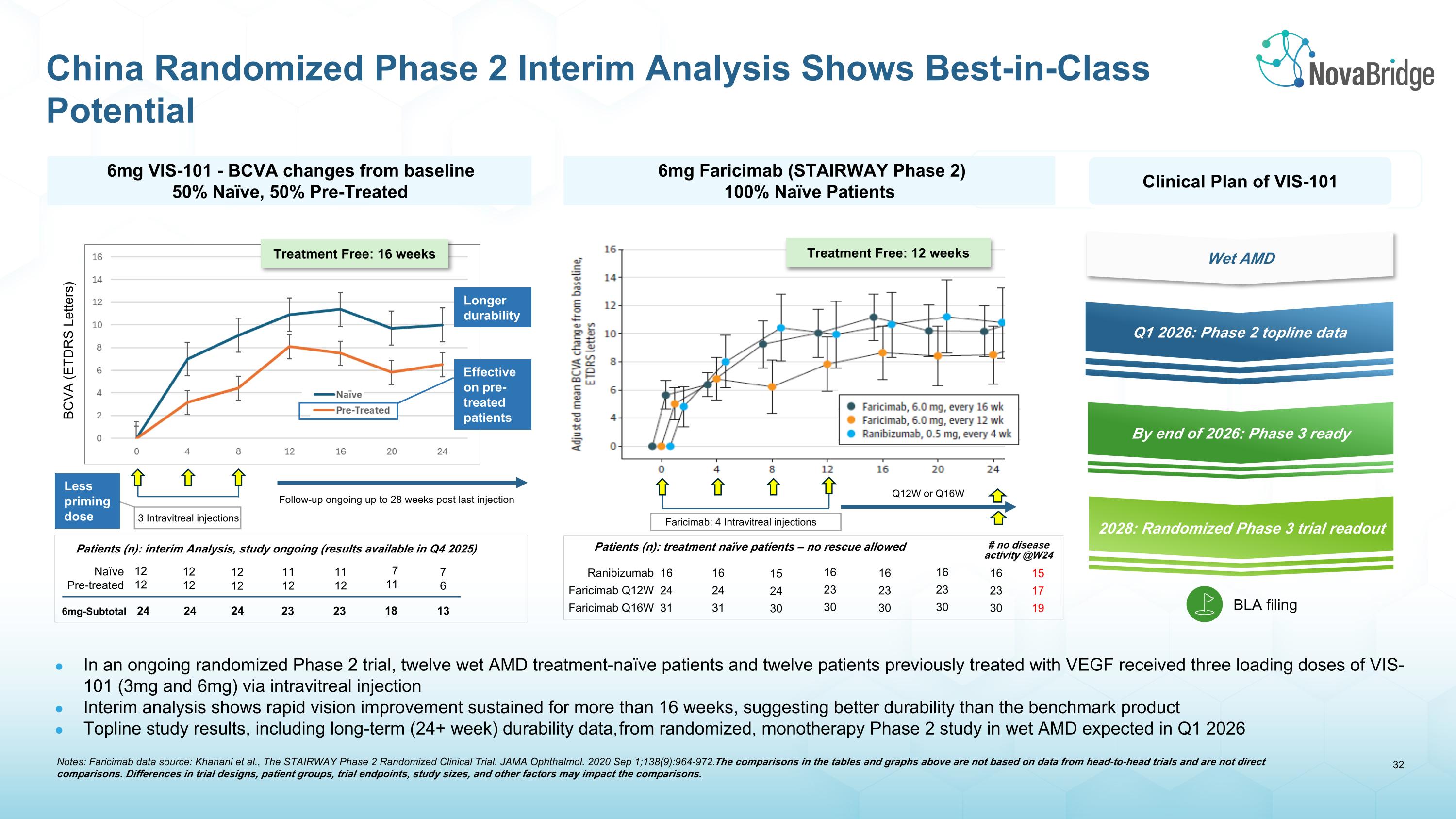
China Randomized Phase 2 Interim Analysis Shows Best-in-Class Potential BCVA (ETDRS Letters) 3 Intravitreal injections Follow-up ongoing up to 28 weeks post last injection Effective on pre-treated patients Less priming dose Longer durability Clinical Plan of VIS-101 Q1 2026: Phase 2 topline data By end of 2026: Phase 3 ready 2028: Randomized Phase 3 trial readout BLA filing Wet AMD 6mg VIS-101 - BCVA changes from baseline 50% Naïve, 50% Pre-Treated In an ongoing randomized Phase 2 trial, twelve wet AMD treatment-naïve patients and twelve patients previously treated with VEGF received three loading doses of VIS-101 (3mg and 6mg) via intravitreal injection Interim analysis shows rapid vision improvement sustained for more than 16 weeks, suggesting better durability than the benchmark product Topline study results, including long-term (24+ week) durability data, from randomized, monotherapy Phase 2 study in wet AMD expected in Q1 2026 Treatment Free: 16 weeks Faricimab: 4 Intravitreal injections Q12W or Q16W Treatment Free: 12 weeks Naïve Pre-treated 12 12 12 12 12 12 11 12 11 12 7 11 7 6 Patients (n): interim Analysis, study ongoing (results available in Q4 2025) 6mg-Subtotal 24 24 24 23 23 18 13 6mg Faricimab (STAIRWAY Phase 2) 100% Naïve Patients Ranibizumab Faricimab Q12W Faricimab Q16W 16 24 31 16 24 31 15 24 30 16 23 30 16 23 30 16 23 30 16 23 30 Patients (n): treatment naïve patients – no rescue allowed # no disease activity @W24 15 17 19 Notes: Faricimab data source: Khanani et al., The STAIRWAY Phase 2 Randomized Clinical Trial. JAMA Ophthalmol. 2020 Sep 1;138(9):964-972. The comparisons in the tables and graphs above are not based on data from head-to-head trials and are not direct comparisons. Differences in trial designs, patient groups, trial endpoints, study sizes, and other factors may impact the comparisons.

Financial Overview�and Upcoming Catalysts
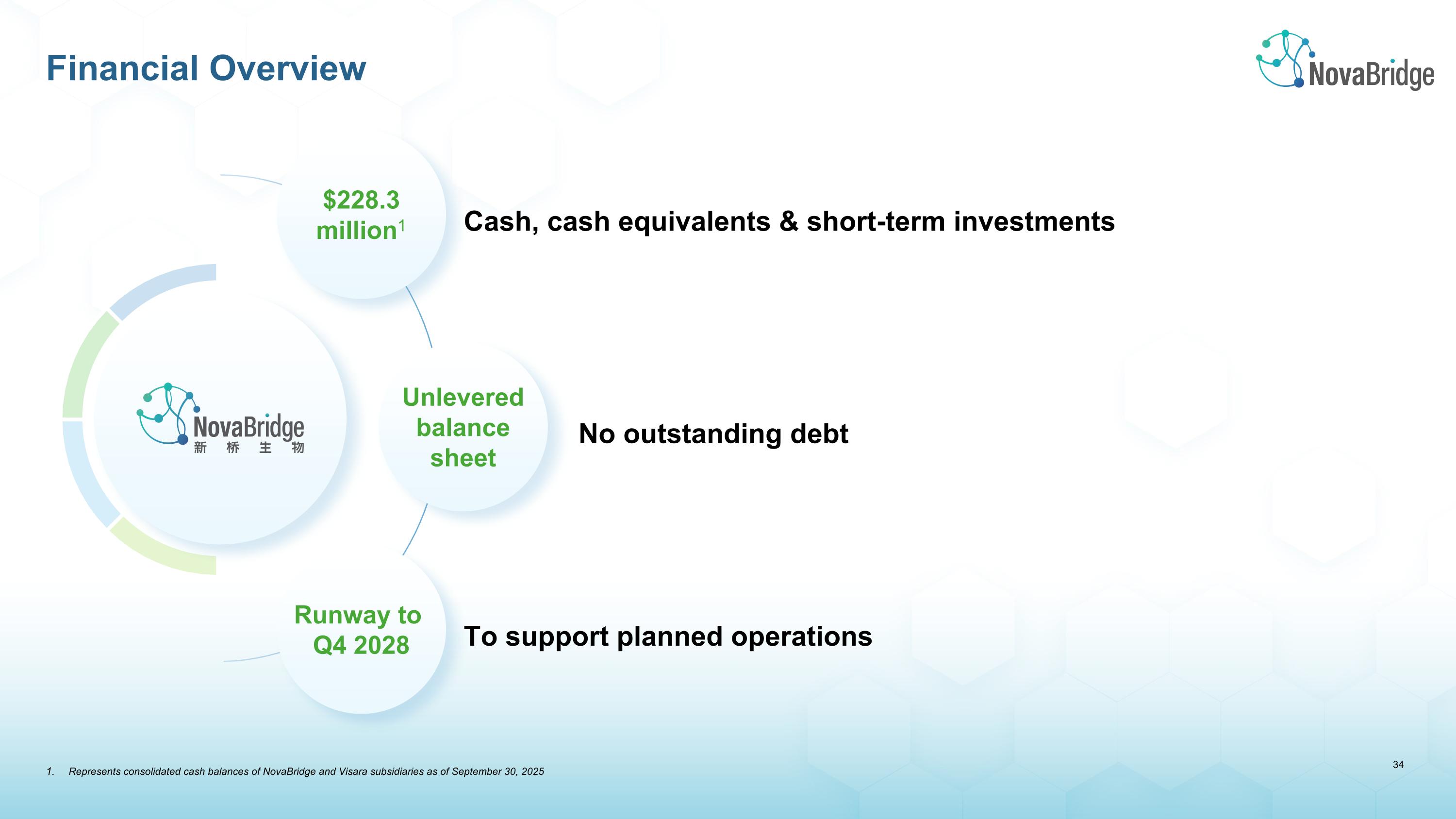
Financial Overview No outstanding debt Cash, cash equivalents & short-term investments To support planned operations $228.3 million1 Runway to �Q4 2028 Unlevered�balance�sheet Represents consolidated cash balances of NovaBridge and Visara subsidiaries as of September 30, 2025
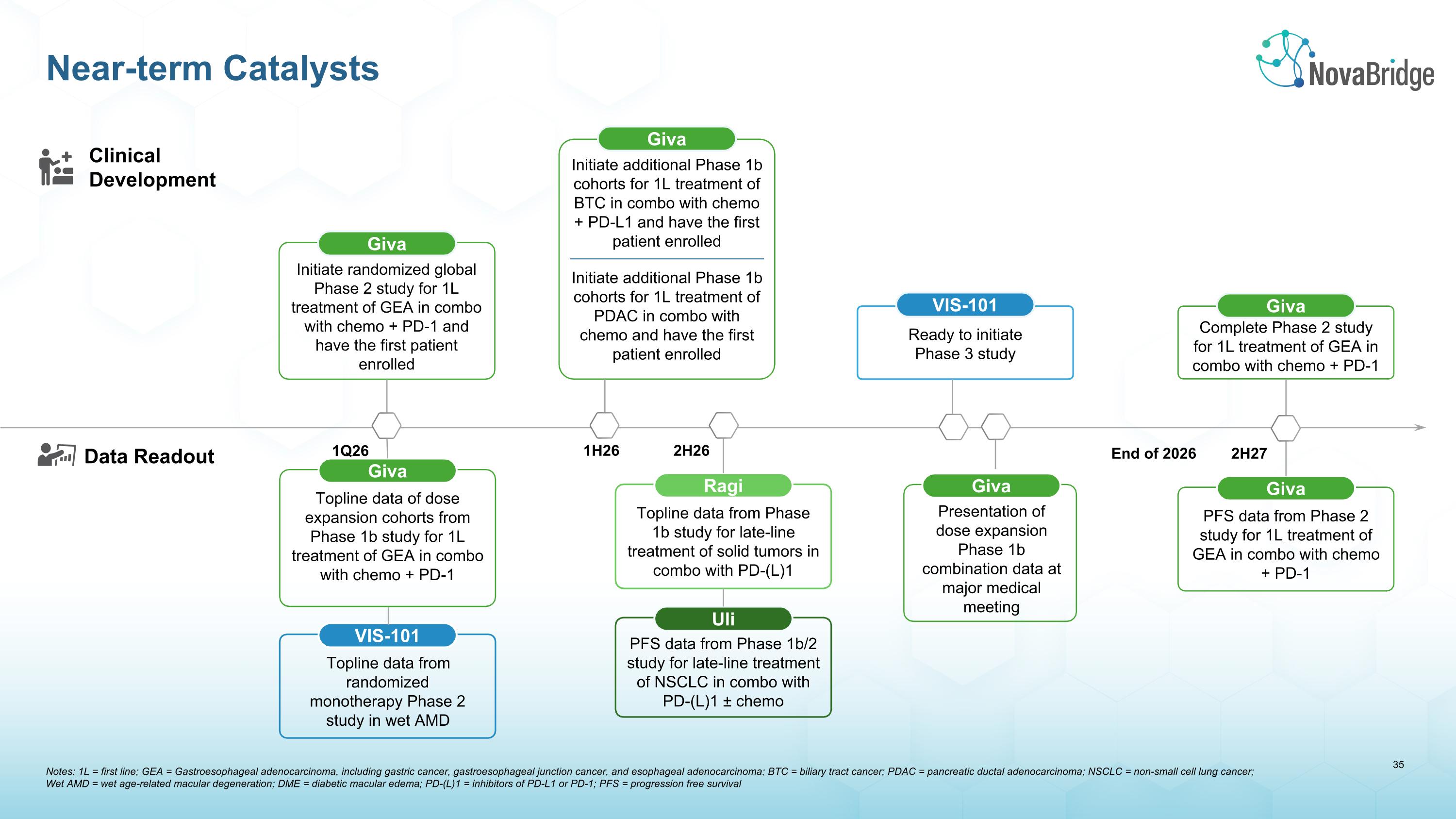
Notes: 1L = first line; GEA = Gastroesophageal adenocarcinoma, including gastric cancer, gastroesophageal junction cancer, and esophageal adenocarcinoma; BTC = biliary tract cancer; PDAC = pancreatic ductal adenocarcinoma; NSCLC = non-small cell lung cancer; �Wet AMD = wet age-related macular degeneration; DME = diabetic macular edema; PD-(L)1 = inhibitors of PD-L1 or PD-1; PFS = progression free survival Near-term Catalysts Clinical�Development Data Readout VIS-101 Topline data from randomized monotherapy Phase 2 study in wet AMD 2H26 Ragi Topline data from Phase 1b study for late-line treatment of solid tumors in combo with PD-(L)1 Uli PFS data from Phase 1b/2 study for late-line treatment of NSCLC in combo with PD-(L)1 ± chemo 1Q26 Giva Topline data of dose expansion cohorts from Phase 1b study for 1L treatment of GEA in combo with chemo + PD-1 2H27 Giva PFS data from Phase 2 study for 1L treatment of GEA in combo with chemo + PD-1 Giva Complete Phase 2 study for 1L treatment of GEA in combo with chemo + PD-1 End of 2026 Ready to initiate Phase 3 study VIS-101 Giva Initiate randomized global Phase 2 study for 1L treatment of GEA in combo with chemo + PD-1 and have the first patient enrolled 1H26 Giva Initiate additional Phase 1b cohorts for 1L treatment of PDAC in combo with chemo and have the first patient enrolled Initiate additional Phase 1b cohorts for 1L treatment of BTC in combo with chemo + PD-L1 and have the first patient enrolled Giva Presentation of dose expansion Phase 1b combination data at major medical meeting

Appendix

Applications pending, expiration date approximated based on expected grant date(s) Intellectual Property Portfolio for Givastomig 2037 2038 2039 2040 2041 2042 2043 2044 2045 2046 US 17/286,437 (Granted) Composition of Matter / Method of Use Treatment of Cancer Aug-2040 US 18/866,687 (Pending)1 Method of Use Dosing Regimen May-20431 PCT/CN2023/122092 (Pending)1 Method of Use Combination with Second Therapeutic Agent Sep-20431 US 63/693,641 (Pending)1 Method of Use Dosing Regimen and Combination Therapy Sep-20451 Treatment of Cancer Dosing Regimen Combination with Second Therapeutic Agent Dosing Regimen and Combination Therapy

Other Oncology Programs Ragistomig Uliledlimab
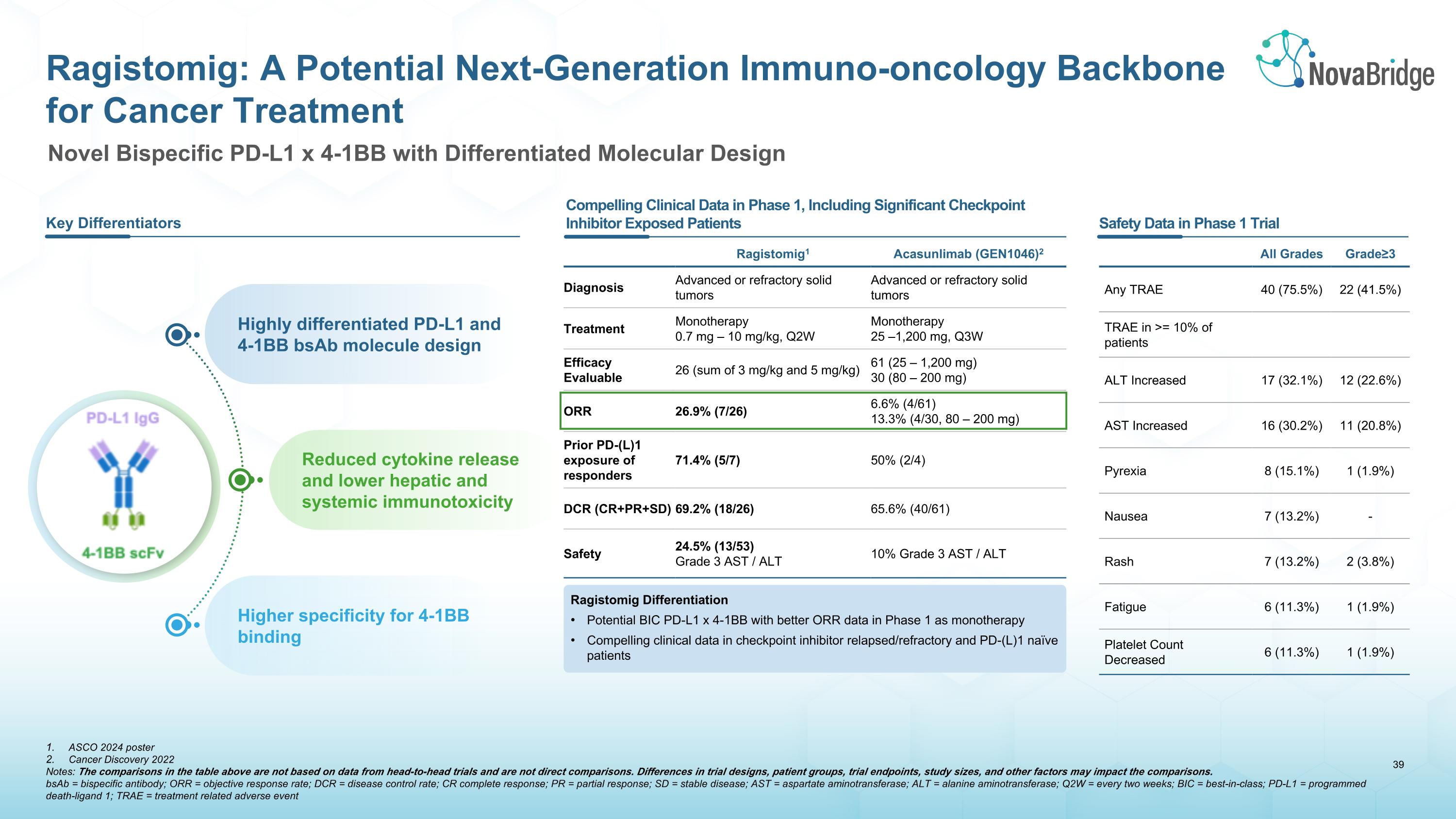
ASCO 2024 poster Cancer Discovery 2022 Notes: The comparisons in the table above are not based on data from head-to-head trials and are not direct comparisons. Differences in trial designs, patient groups, trial endpoints, study sizes, and other factors may impact the comparisons. bsAb = bispecific antibody; ORR = objective response rate; DCR = disease control rate; CR complete response; PR = partial response; SD = stable disease; AST = aspartate aminotransferase; ALT = alanine aminotransferase; Q2W = every two weeks; BIC = best-in-class; PD-L1 = programmed death-ligand 1; TRAE = treatment related adverse event Ragistomig: A Potential Next-Generation Immuno-oncology Backbone for Cancer Treatment Key Differentiators Highly differentiated PD-L1 and 4-1BB bsAb molecule design Reduced cytokine release and lower hepatic and systemic immunotoxicity Higher specificity for 4-1BB binding Compelling Clinical Data in Phase 1, Including Significant Checkpoint Inhibitor Exposed Patients Safety Data in Phase 1 Trial Ragistomig1 Acasunlimab (GEN1046)2 Diagnosis Advanced or refractory solid tumors Advanced or refractory solid tumors Treatment Monotherapy�0.7 mg – 10 mg/kg, Q2W Monotherapy�25 –1,200 mg, Q3W Efficacy �Evaluable 26 (sum of 3 mg/kg and 5 mg/kg) 61 (25 – 1,200 mg)�30 (80 – 200 mg) ORR 26.9% (7/26) 6.6% (4/61)�13.3% (4/30, 80 – 200 mg) Prior PD-(L)1 exposure of responders 71.4% (5/7) 50% (2/4) DCR (CR+PR+SD) 69.2% (18/26) 65.6% (40/61) Safety 24.5% (13/53)�Grade 3 AST / ALT 10% Grade 3 AST / ALT Ragistomig Differentiation Potential BIC PD-L1 x 4-1BB with better ORR data in Phase 1 as monotherapy Compelling clinical data in checkpoint inhibitor relapsed/refractory and PD-(L)1 naïve patients All Grades Grade≥3 Any TRAE 40 (75.5%) 22 (41.5%) TRAE in >= 10% of patients ALT Increased 17 (32.1%) 12 (22.6%) AST Increased 16 (30.2%) 11 (20.8%) Pyrexia 8 (15.1%) 1 (1.9%) Nausea 7 (13.2%) - Rash 7 (13.2%) 2 (3.8%) Fatigue 6 (11.3%) 1 (1.9%) Platelet Count Decreased 6 (11.3%) 1 (1.9%) Novel Bispecific PD-L1 x 4-1BB with Differentiated Molecular Design
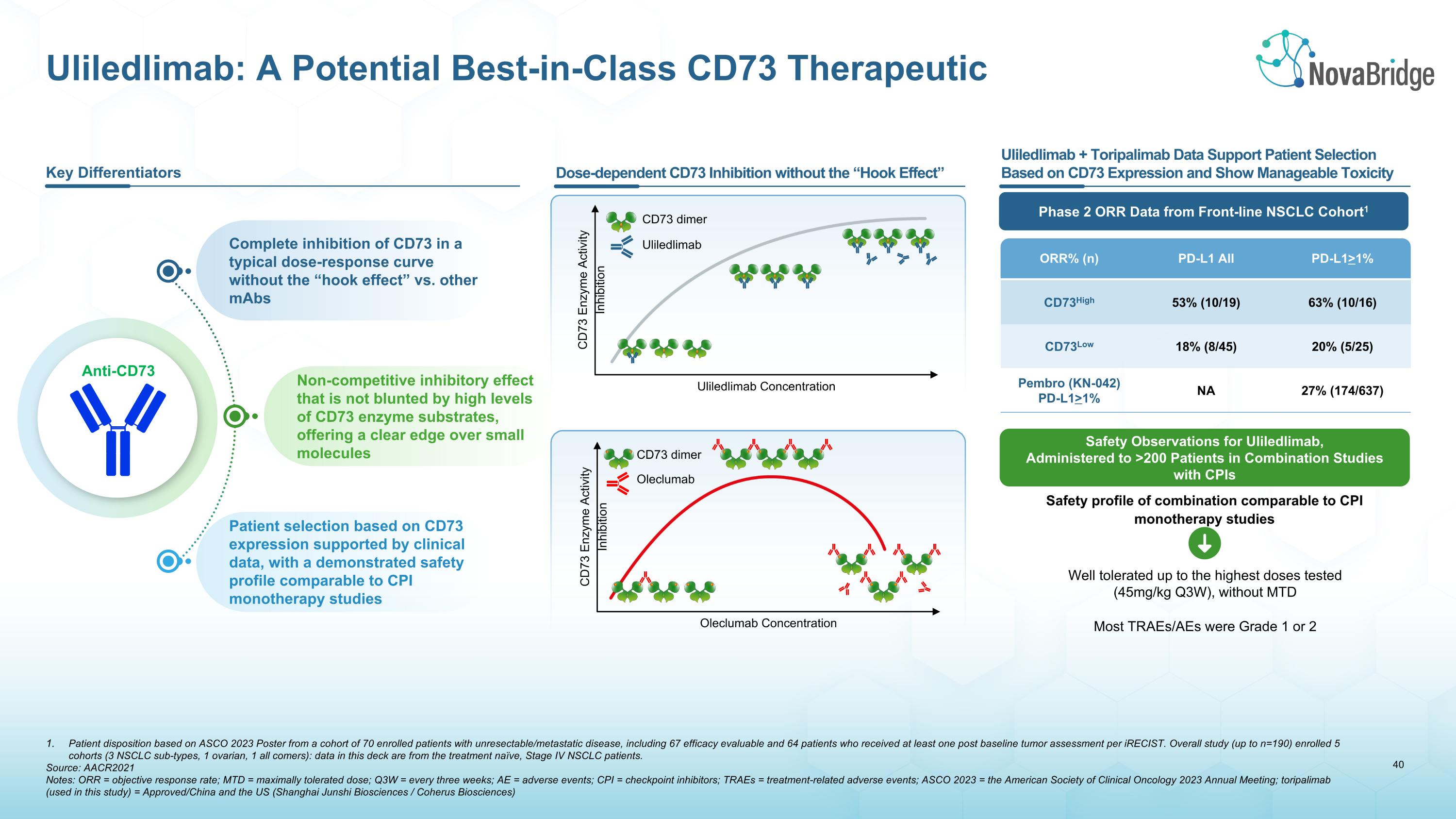
Patient disposition based on ASCO 2023 Poster from a cohort of 70 enrolled patients with unresectable/metastatic disease, including 67 efficacy evaluable and 64 patients who received at least one post baseline tumor assessment per iRECIST. Overall study (up to n=190) enrolled 5 cohorts (3 NSCLC sub-types, 1 ovarian, 1 all comers): data in this deck are from the treatment naïve, Stage IV NSCLC patients. Source: AACR2021 Notes: ORR = objective response rate; MTD = maximally tolerated dose; Q3W = every three weeks; AE = adverse events; CPI = checkpoint inhibitors; TRAEs = treatment-related adverse events; ASCO 2023 = the American Society of Clinical Oncology 2023 Annual Meeting; toripalimab (used in this study) = Approved/China and the US (Shanghai Junshi Biosciences / Coherus Biosciences) Uliledlimab: A Potential Best-in-Class CD73 Therapeutic Key Differentiators Complete inhibition of CD73 in a typical dose-response curve without the “hook effect” vs. other mAbs Non-competitive inhibitory effect that is not blunted by high levels of CD73 enzyme substrates, offering a clear edge over small molecules Patient selection based on CD73 expression supported by clinical data, with a demonstrated safety profile comparable to CPI monotherapy studies Dose-dependent CD73 Inhibition without the “Hook Effect” Uliledlimab + Toripalimab Data Support Patient Selection Based on CD73 Expression and Show Manageable Toxicity CD73 Enzyme Activity Inhibition Uliledlimab Concentration Uliledlimab CD73 dimer CD73 Enzyme Activity Inhibition Oleclumab CD73 dimer Oleclumab Concentration ORR% (n) PD-L1 All PD-L1>1% CD73High 53% (10/19) 63% (10/16) CD73Low 18% (8/45) 20% (5/25) Pembro (KN-042) PD-L1>1% NA 27% (174/637) Phase 2 ORR Data from Front-line NSCLC Cohort1 Safety profile of combination comparable to CPI monotherapy studies Well tolerated up to the highest doses tested �(45mg/kg Q3W), without MTD Most TRAEs/AEs were Grade 1 or 2 Safety Observations for Uliledlimab, Administered to >200 Patients in Combination Studies with CPIs Anti-CD73

Research and Development

Elizabeth Lindner Executive Director, Clinical Operations 10+ Xuejun Liu Executive Director, Translational Medicine 10+ Peter Sabbatini Senior Director, �Clinical Biomarkers 10+ Our Research and Development Capabilities Years of Industry Experience # Claire Xu MD, PhD Senior Vice President, Clinical Development 17 Phillip Dennis MD, PhD Chief Medical Officer 27 Strong R&D Team with Proven Clinical Development Capabilities Extensive and proven clinical development expertise in early- and late-stage oncology drug development Well-positioned to effectively develop compounds through clinical strategies that maximize asset value at the PoC stage Proven Capability to Swiftly Assemble a World-class R&D team 77 Patents under Application 71 Registered Patents Innovative Drug Assets with Global Differentiation Proposition 4 Clinical-stage Programs Achievements Cadmus C. Rich MD, MBA Chief Medical Officer of Visara Established Talent Pool with Outstanding Achievements A broad spectrum of R&D functionalities Capabilities Clinical Development Clinical Biomarkers Translational Medicine Clinical Operations Highly qualified R&D professionals with advanced scientific and medicine degrees 47% of R&D Members hold Doctorate Degree Deep and extensive experience in drug development Over 10 years of industry experience for each R&D member Rapidly-assembled world-class team of Visara Compelling opportunity of assets attracting seasoned leaders Strong commitment to build a world-class organization Extensive industry resources backed by CBC Group Key Drivers Recent Appointment of Distinguished Ophthalmology Leaders for Visara Carlos Quezada-Ruiz MD Chair of the Scientific Advisory Board of Visara Emmett Cunningham MD, PhD Co-Founder and Executive Chairman of Visara

Strategic Advantages

Asia’s largest and most impactful healthcare-dedicated asset manager Investor-Operator approach Build deep expertise in value-creation levers and hands-on portfolio management, securing significantly greater exposures to top-tier assets compared with peers CBC is well positioned with global resources and validated recipe for fast scaling healthcare companies Partnering with the world's top scientists and entrepreneurs, CBC's unique investor-operator approach has empowered leading global healthcare companies to widen access to quality and affordable medical care, catalyze innovations worldwide, and provide better healthcare for all AUM US$10.5b, Buy-out investment US$3.5b One of the largest and most dynamic healthcare ecosystems across all major healthcare verticals: 30+ healthcare portfolio companies , 20+ controlled healthcare companies, 40+ innovative drug pipeline assets and 20+ early-stage pipeline assets 40+ BD deals within the last 5 years with 20+ global MNCs and partners with assets at different stages of development and transaction types Global talent pool with 100+ senior operating talents, 2,000+ candidates and 1,000+ experts available Proper governance Learn together with management while ensuring proper governance to drive the right behaviors and manage risk Talent warehousing Build and maintain a robust network of experienced science / operating partners Extensive global network and coverage Leverage dynamic healthcare ecosystems to identify high potential assets and valuable investment targets worldwide Intra-Portfolio integration Maximize synergies within expansive portfolio and consolidate at arm’s length manner CBC is a Steadfast Investor-Operator Differentiating from Peers

Regional Coverage Leads Sean LU Senior Managing Director Greater China Billy CHO Senior Managing Director Korea Vijay KARWAL Managing Director SEA Global Investment Team Managing Directors Directors / Vice Presidents Neo ZHANG Managing Director Hao YIN Managing Director Ray JIN Managing Director Qiuyi LIU Director Sangsoo KIM Director Sam LIAO Director Henry QU Vice President Mars LIN Director Paul QI Vice President Randy YEO Vice President Harry SUN Managing Director Denny CHU Managing Director US Japan Local-for-Local Approach with Centralized Underwriting Support Highly Experienced World-Class Team to Support Global Strategy
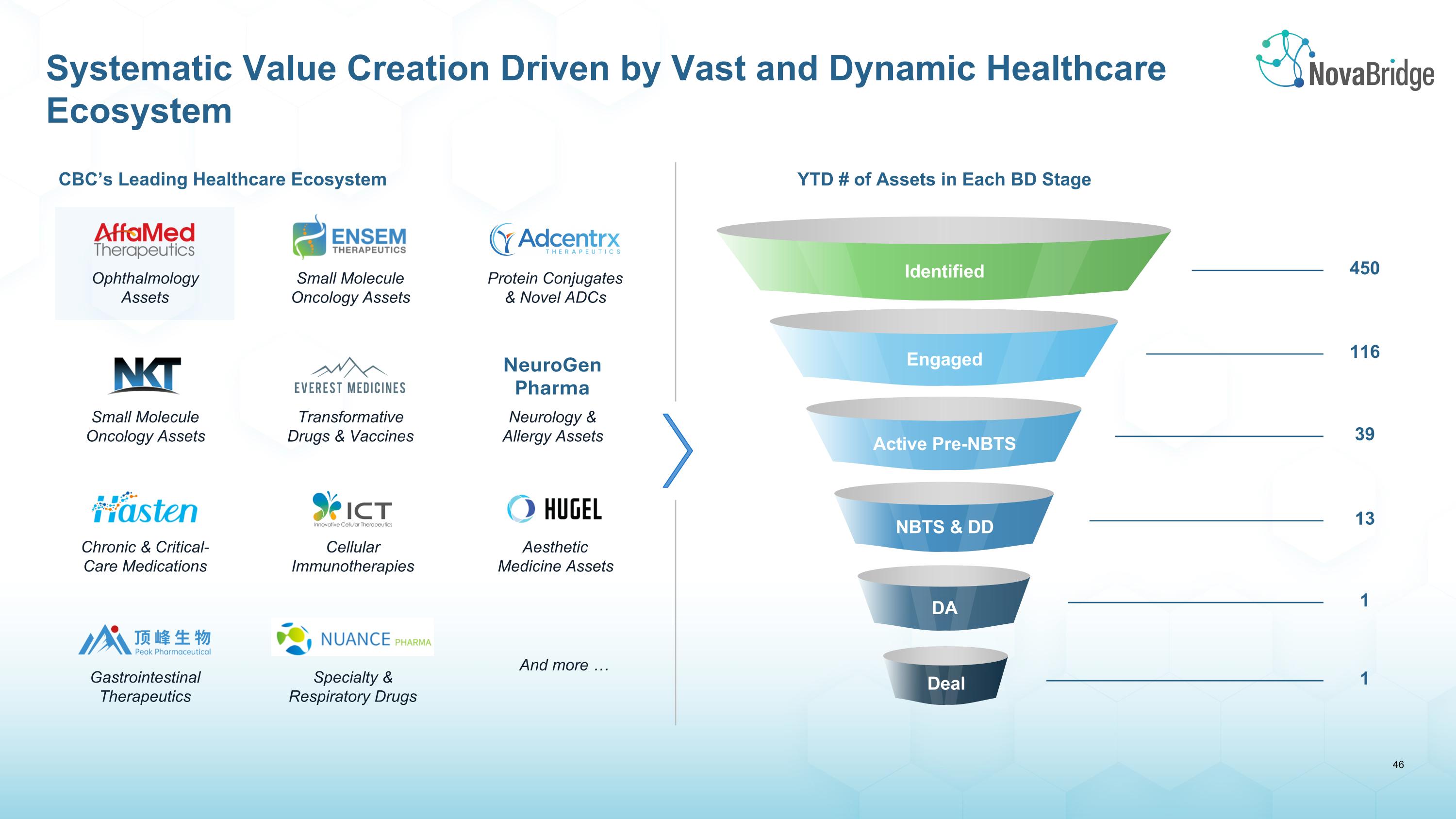
Ophthalmology Assets Small Molecule Oncology Assets Protein Conjugates & Novel ADCs Small Molecule Oncology Assets Transformative Drugs & Vaccines Neurology & Allergy Assets NeuroGen Pharma And more … Gastrointestinal Therapeutics Specialty & Respiratory Drugs Chronic & Critical-Care Medications Cellular Immunotherapies Aesthetic Medicine Assets CBC’s Leading Healthcare Ecosystem YTD # of Assets in Each BD Stage Identified Engaged 450 116 Active Pre-NBTS 39 NBTS & DD 13 DA 1 1 Deal Systematic Value Creation Driven by Vast and Dynamic Healthcare Ecosystem

Collaborations

Collaboration Arrangements: Collaboration with ABL Bio for Givastomig and Ragistomig ABL Bio is a research-led biotechnology company and a pioneer in bispecific antibodies for immuno-oncology and neurodegenerative diseases For givastomig, NovaBridge acts as the Lead Party worldwide excluding South Korea and Greater China (ROW, which includes the United States). For ragistomig, ABL Bio acts as the Lead Party in the ROW The Lead Party has the right to (i) make decisions regarding a drug candidate’s development plans in the ROW, and (ii) make final decisions to enter into out-licensing agreements with respect to a drug candidate in any country in the ROW Givastomig (Lead Party: ) Ragistomig (Lead Party: ) Development & Commercialization Rights ROW: NovaBridge to make final decisions regarding development plans and out-licensing agreements, NovaBridge will be the MAH South Korea: Handok has the exclusive rights Greater China: TJ Bio (Shanghai) has the exclusive rights ROW: ABL Bio to make final decisions regarding development plans and out-licensing agreements, ABL Bio will be the MAH South Korea and Greater China: ABL Bio has the exclusive rights Intellectual Property Rights Intellectual property rights in the ROW shall be jointly and equally owned by NovaBridge and ABL Bio Income Sharing & Development Cost-Sharing The income derived from out-licensing agreement and development cost will be 50-50 shared by NovaBridge and ABL Bio

The Visara Territory refers to all countries and territories worldwide except for Singapore, Thailand, Malaysia, Indonesia, Vietnam, the People’s Republic of China, Taiwan, Macau, Hong Kong, Korea, and India Certain Territories refers to Singapore, Thailand, Malaysia, Indonesia, Vietnam, the People’s Republic of China, Taiwan, Macau, Hong Kong, Korea, and India Collaboration Arrangements: The In-License and Subsequent Assignment of VIS-101 In-license of VIS-101 in Visara Territory1 Visara, Inc. NovaBridge AffaMed ESOP 65% 30% 5% In-license and Subsequent Assignment of VIS-101 in Certain Territories2 Visara entered into an Exclusive License Agreement with AskGene, pursuant to which AskGene grants Visara an exclusive, royalty bearing license, with the right to sublicense, develop, commercialize and otherwise exploit the licensed product (including VIS-101) in Certain Territories In-license Subsequent Assignment Visara entered into an Assignment and Assumption Agreement with Everest Medicines, pursuant to which Visara assigns all of its rights and obligations under the License Agreement with AskGene to Everest Medicines, including the exclusive, royalty bearing license to develop, commercialize and otherwise exploit VIS-101 in Certain Territories NovaBridge contributed cash in exchange for 65% of the equity interest in Visara AffaMed contributed its rights and interests in VIS-101 to Visara in exchange for 30% of the equity interest in Visara The remaining 5% equity interest in Visara reserved for an ESOP VIS-101-related ownership rights are shown above

Thank you www.novabridge.com IR@novabridge.com




















































